Mechanisms regulating neuronal excitability and seizure development following mTOR pathway hyperactivation
- PMID: 24672426
- PMCID: PMC3953715
- DOI: 10.3389/fnmol.2014.00018
Mechanisms regulating neuronal excitability and seizure development following mTOR pathway hyperactivation
Abstract
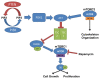
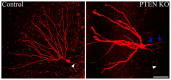
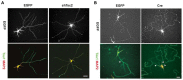
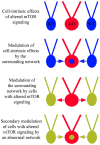
The phosphatidylinositol-3-kinase/phosphatase and tensin homolog (PTEN)-mammalian target of rapamycin (mTOR) pathway regulates a variety of neuronal functions, including cell proliferation, survival, growth, and plasticity. Dysregulation of the pathway is implicated in the development of both genetic and acquired epilepsies. Indeed, several causal mutations have been identified in patients with epilepsy, the most prominent of these being mutations in PTEN and tuberous sclerosis complexes 1 and 2 (TSC1, TSC2). These genes act as negative regulators of mTOR signaling, and mutations lead to hyperactivation of the pathway. Animal models deleting PTEN, TSC1, and TSC2 consistently produce epilepsy phenotypes, demonstrating that increased mTOR signaling can provoke neuronal hyperexcitability. Given the broad range of changes induced by altered mTOR signaling, however, the mechanisms underlying seizure development in these animals remain uncertain. In transgenic mice, cell populations with hyperactive mTOR have many structural abnormalities that support recurrent circuit formation, including somatic and dendritic hypertrophy, aberrant basal dendrites, and enlargement of axon tracts. At the functional level, mTOR hyperactivation is commonly, but not always, associated with enhanced synaptic transmission and plasticity. Moreover, these populations of abnormal neurons can affect the larger network, inducing secondary changes that may explain paradoxical findings reported between cell and network functioning in different models or at different developmental time points. Here, we review the animal literature examining the link between mTOR hyperactivation and epileptogenesis, emphasizing the impact of enhanced mTOR signaling on neuronal form and function.
Keywords: PTEN; TSC; autism; epilepsy; granule cells; hippocampus; mTOR; neurogenesis.
Figures


Similar articles
-
A circuitry and biochemical basis for tuberous sclerosis symptoms: from epilepsy to neurocognitive deficits.Int J Dev Neurosci. 2013 Nov;31(7):667-78. doi: 10.1016/j.ijdevneu.2013.02.008. Epub 2013 Feb 26. Int J Dev Neurosci. 2013. PMID: 23485365 Free PMC article. Review.
-
Finding a better drug for epilepsy: the mTOR pathway as an antiepileptogenic target.Epilepsia. 2012 Jul;53(7):1119-30. doi: 10.1111/j.1528-1167.2012.03506.x. Epub 2012 May 11. Epilepsia. 2012. PMID: 22578218 Free PMC article. Review.
-
Mechanistic target of rapamycin (mTOR) in tuberous sclerosis complex-associated epilepsy.Pediatr Neurol. 2015 Mar;52(3):281-9. doi: 10.1016/j.pediatrneurol.2014.10.028. Epub 2014 Nov 20. Pediatr Neurol. 2015. PMID: 25591831 Review.
-
Mammalian target of rapamycin inhibitors for treatment in tuberous sclerosis.Korean J Pediatr. 2011 Jun;54(6):241-5. doi: 10.3345/kjp.2011.54.6.241. Epub 2011 Jun 30. Korean J Pediatr. 2011. Retraction in: Korean J Pediatr. 2013 Jun;56(6):269. doi: 10.3345/kjp.2013.56.6.269. PMID: 21949518 Free PMC article. Retracted.
-
Biallelic Mutations in TSC2 Lead to Abnormalities Associated with Cortical Tubers in Human iPSC-Derived Neurons.J Neurosci. 2019 Nov 20;39(47):9294-9305. doi: 10.1523/JNEUROSCI.0642-19.2019. Epub 2019 Oct 7. J Neurosci. 2019. PMID: 31591157 Free PMC article.
Cited by
-
Current Review in Basic Science: Animal Models of Focal Cortical Dysplasia and Epilepsy.Epilepsy Curr. 2022 Apr 22;22(4):234-240. doi: 10.1177/15357597221098230. eCollection 2022 Jul-Aug. Epilepsy Curr. 2022. PMID: 36187145 Free PMC article. Review.
-
Molecular Mechanism and Regulation of Autophagy and Its Potential Role in Epilepsy.Cells. 2022 Aug 23;11(17):2621. doi: 10.3390/cells11172621. Cells. 2022. PMID: 36078029 Free PMC article. Review.
-
Epileptogenesis in tuberous sclerosis complex-related developmental and epileptic encephalopathy.Brain. 2023 Jul 3;146(7):2694-2710. doi: 10.1093/brain/awad048. Brain. 2023. PMID: 36806388 Free PMC article. Review.
-
New clinicopathological associations and histoprognostic markers in ILAE types of hippocampal sclerosis.Brain Pathol. 2018 Sep;28(5):644-655. doi: 10.1111/bpa.12596. Epub 2018 Apr 10. Brain Pathol. 2018. PMID: 29476662 Free PMC article.
-
Seizure control through genetic and pharmacological manipulation of Pumilio in Drosophila: a key component of neuronal homeostasis.Dis Model Mech. 2017 Feb 1;10(2):141-150. doi: 10.1242/dmm.027045. Epub 2016 Dec 14. Dis Model Mech. 2017. PMID: 28067623 Free PMC article.
References
-
- Arch E. M., Goodman B. K., Van Wesep R. A., Liaw D., Clarke K., Parsons R., et al. (1997). Deletion of PTEN in a patient with Bannayan–Riley–Ruvalcaba syndrome suggests allelism with Cowden disease. Am. J. Med. Genet. 71 489–493 10.1002/(SICI)1096-8628(19970905)71:4<489::AID-AJMG24>3.0.CO;2-B - DOI - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

