Defective autophagy in spastizin mutated patients with hereditary spastic paraparesis type 15
- PMID: 24030950
- PMCID: PMC3784282
- DOI: 10.1093/brain/awt227
Defective autophagy in spastizin mutated patients with hereditary spastic paraparesis type 15
Abstract
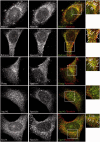
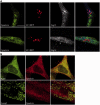
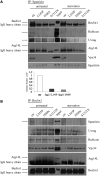
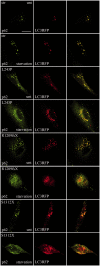
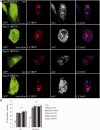
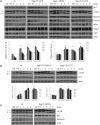
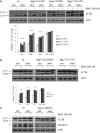
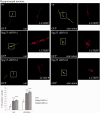
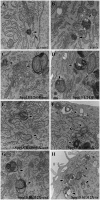
Hereditary spastic paraparesis type 15 is a recessive complicated form of the disease clinically characterized by slowly progressive spastic paraparesis and mental deterioration with onset between the first and second decade of life. Thinning of corpus callosum is the neuroradiological distinctive sign frequently associated with white matter abnormalities. The causative gene, ZFYVE26, encodes a large protein of 2539 amino acid residues, termed spastizin, containing three recognizable domains: a zinc finger, a leucine zipper and a FYVE domain. Spastizin protein has a diffuse cytoplasmic distribution and co-localizes partially with early endosomes, the endoplasmic reticulum, microtubules and vesicles involved in protein trafficking. In addition, spastizin localizes to the mid-body during the final step of mitosis and contributes to successful cytokinesis. Spastizin interacts with Beclin 1, a protein required for cytokinesis and autophagy, which is the major lysosome-mediated degradation process in the cell. In view of the Beclin 1-spastizin interaction, we investigated the possible role of spastizin in autophagy. We carried out this analysis by using lymphoblast and fibroblast cells derived from four different spastizin mutated patients (p.I508N, p.L243P, p.R1209fsX, p.S1312X) and from control subjects. Of note, the truncating p.R1209fsX and p.S1312X mutations lead to loss of spastizin protein. The results obtained indicate that spastizin interacts with the autophagy related Beclin 1-UVRAG-Rubicon multiprotein complex and is required for autophagosome maturation. In cells lacking spastizin or with mutated forms of the protein, spastizin interaction with Beclin 1 is lost although the formation of the Beclin 1-UVRAG-Rubicon complex can still be observed. However, in these cells we demonstrate an impairment of autophagosome maturation and an accumulation of immature autophagosomes. Autophagy defects with autophagosome accumulation can be observed also in neuronal cells upon spastizin silencing. These results indicate that autophagy is a central process in the pathogenesis of complicated forms of hereditary spastic paraparesis with thin corpus callosum.
Keywords: Beclin 1; SPG15; autophagosome maturation; autophagy; spastizin.
Figures



Similar articles
-
ZFYVE26/SPASTIZIN and SPG11/SPATACSIN mutations in hereditary spastic paraplegia types AR-SPG15 and AR-SPG11 have different effects on autophagy and endocytosis.Autophagy. 2019 Jan;15(1):34-57. doi: 10.1080/15548627.2018.1507438. Epub 2018 Sep 13. Autophagy. 2019. PMID: 30081747 Free PMC article.
-
A hereditary spastic paraplegia mouse model supports a role of ZFYVE26/SPASTIZIN for the endolysosomal system.PLoS Genet. 2013;9(12):e1003988. doi: 10.1371/journal.pgen.1003988. Epub 2013 Dec 19. PLoS Genet. 2013. PMID: 24367272 Free PMC article.
-
ZFYVE26/SPASTIZIN: a close link between complicated hereditary spastic paraparesis and autophagy.Autophagy. 2014 Feb;10(2):374-5. doi: 10.4161/auto.27173. Epub 2013 Nov 26. Autophagy. 2014. PMID: 24284334 Free PMC article.
-
Impaired autophagy and APP processing in Alzheimer's disease: The potential role of Beclin 1 interactome.Prog Neurobiol. 2013 Jul-Aug;106-107:33-54. doi: 10.1016/j.pneurobio.2013.06.002. Epub 2013 Jul 1. Prog Neurobiol. 2013. PMID: 23827971 Review.
-
Congenital disorders of autophagy: an emerging novel class of inborn errors of neuro-metabolism.Brain. 2016 Feb;139(Pt 2):317-37. doi: 10.1093/brain/awv371. Epub 2015 Dec 29. Brain. 2016. PMID: 26715604 Free PMC article. Review.
Cited by
-
Loss of the Mitochondrial Fission GTPase Drp1 Contributes to Neurodegeneration in a Drosophila Model of Hereditary Spastic Paraplegia.Brain Sci. 2020 Sep 17;10(9):646. doi: 10.3390/brainsci10090646. Brain Sci. 2020. PMID: 32957716 Free PMC article.
-
The different autophagy degradation pathways and neurodegeneration.Neuron. 2022 Mar 16;110(6):935-966. doi: 10.1016/j.neuron.2022.01.017. Epub 2022 Feb 7. Neuron. 2022. PMID: 35134347 Free PMC article. Review.
-
Adult-Onset Hereditary Spastic Paraplegia 15 in a Saudi Patient with A Compound Heterozygous Variant in the ZFYVE26Gene.Maedica (Bucur). 2022 Sep;17(3):730-734. doi: 10.26574/maedica.2022.17.3.730. Maedica (Bucur). 2022. PMID: 36540605 Free PMC article.
-
Autophagy and human diseases.Cell Res. 2014 Jan;24(1):69-79. doi: 10.1038/cr.2013.161. Epub 2013 Dec 10. Cell Res. 2014. PMID: 24323045 Free PMC article. Review.
-
Axon demyelination and degeneration in a zebrafish spastizin model of hereditary spastic paraplegia.Open Biol. 2024 Nov;14(11):240100. doi: 10.1098/rsob.240100. Epub 2024 Nov 6. Open Biol. 2024. PMID: 39503232 Free PMC article.
References
-
- Boland B, Nixon RA. Neuronal macroautophagy: from development to degeneration. Mol Aspects Med. 2006;27:503–19. - PubMed
-
- Boukhris A, Stevanin G, Feki I, Denora P, Elleuch N, Miladi MI, et al. Tunisian hereditary spastic paraplegias: clinical variability supported by genetic heterogeneity. Clin Genet. 2009;75:527–36. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

