Comprehensive clinical and molecular analysis of 12 families with type 1 recessive cutis laxa
- PMID: 22829427
- PMCID: PMC4105850
- DOI: 10.1002/humu.22165
Comprehensive clinical and molecular analysis of 12 families with type 1 recessive cutis laxa
Abstract
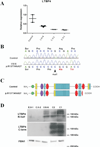
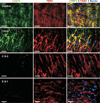
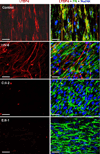
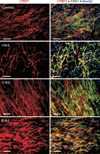
Autosomal recessive cutis laxa type I (ARCL type I) is characterized by generalized cutis laxa with pulmonary emphysema and/or vascular complications. Rarely, mutations can be identified in FBLN4 or FBLN5. Recently, LTBP4 mutations have been implicated in a similar phenotype. Studying FBLN4, FBLN5, and LTBP4 in 12 families with ARCL type I, we found bi-allelic FBLN5 mutations in two probands, whereas nine probands harbored biallelic mutations in LTBP4. FBLN5 and LTBP4 mutations cause a very similar phenotype associated with severe pulmonary emphysema, in the absence of vascular tortuosity or aneurysms. Gastrointestinal and genitourinary tract involvement seems to be more severe in patients with LTBP4 mutations. Functional studies showed that most premature termination mutations in LTBP4 result in severely reduced mRNA and protein levels. This correlated with increased transforming growth factor-beta (TGFβ) activity. However, one mutation, c.4127dupC, escaped nonsense-mediated decay. The corresponding mutant protein (p.Arg1377Alafs(*) 27) showed reduced colocalization with fibronectin, leading to an abnormal morphology of microfibrils in fibroblast cultures, while retaining normal TGFβ activity. We conclude that LTBP4 mutations cause disease through both loss of function and gain of function mechanisms.
© 2012 Wiley Periodicals, Inc.
Conflict of interest statement
The authors have no conflict of interest to declare.
Figures

Similar articles
-
LTBP4 in Health and Disease.Genes (Basel). 2021 May 23;12(6):795. doi: 10.3390/genes12060795. Genes (Basel). 2021. PMID: 34071145 Free PMC article. Review.
-
Clinical and molecular characterization of an 18-month-old infant with autosomal recessive cutis laxa type 1C due to a novel LTBP4 pathogenic variant, and literature review.Mol Genet Genomic Med. 2019 Jul;7(7):e00735. doi: 10.1002/mgg3.735. Epub 2019 May 21. Mol Genet Genomic Med. 2019. PMID: 31115174 Free PMC article. Review.
-
Two novel compound heterozygous variants of LTBP4 in a Chinese infant with cutis laxa type IC and a review of the related literature.BMC Med Genomics. 2020 Dec 10;13(1):183. doi: 10.1186/s12920-020-00842-6. BMC Med Genomics. 2020. PMID: 33302946 Free PMC article. Review.
-
Autosomal recessive cutis laxa: a novel mutation in the FBLN5 gene in a family.Clin Dysmorphol. 2019 Apr;28(2):63-65. doi: 10.1097/MCD.0000000000000258. Clin Dysmorphol. 2019. PMID: 30640789
-
Latent transforming growth factor binding protein 4 regulates transforming growth factor beta receptor stability.Hum Mol Genet. 2015 Jul 15;24(14):4024-36. doi: 10.1093/hmg/ddv139. Epub 2015 Apr 16. Hum Mol Genet. 2015. PMID: 25882708 Free PMC article.
Cited by
-
Defining the Clinical, Molecular and Ultrastructural Characteristics in Occipital Horn Syndrome: Two New Cases and Review of the Literature.Genes (Basel). 2019 Jul 12;10(7):528. doi: 10.3390/genes10070528. Genes (Basel). 2019. PMID: 31336972 Free PMC article. Review.
-
Fibrillin microfibrils and elastic fibre proteins: Functional interactions and extracellular regulation of growth factors.Semin Cell Dev Biol. 2019 May;89:109-117. doi: 10.1016/j.semcdb.2018.07.016. Epub 2018 Jul 20. Semin Cell Dev Biol. 2019. PMID: 30016650 Free PMC article. Review.
-
Extracellular matrix in lung development, homeostasis and disease.Matrix Biol. 2018 Nov;73:77-104. doi: 10.1016/j.matbio.2018.03.005. Epub 2018 Mar 8. Matrix Biol. 2018. PMID: 29524630 Free PMC article. Review.
-
LTBP4 in Health and Disease.Genes (Basel). 2021 May 23;12(6):795. doi: 10.3390/genes12060795. Genes (Basel). 2021. PMID: 34071145 Free PMC article. Review.
-
Cutis laxa: intersection of elastic fiber biogenesis, TGFβ signaling, the secretory pathway and metabolism.Matrix Biol. 2014 Jan;33:16-22. doi: 10.1016/j.matbio.2013.07.006. Epub 2013 Aug 16. Matrix Biol. 2014. PMID: 23954411 Free PMC article. Review.
References
-
- Abe M, Harpel JG, Metz CN, Nunes I, Loskutoff DJ, Rifkin DB. An assay for transforming growth factor-beta using cells transfected with a plasminogen activator inhibitor-1 promoter-luciferase construct. Anal Biochem. 1994;216(2):276–284. - PubMed
-
- Bicknell LS, Pitt J, Aftimos S, Ramadas R, Maw MA, Robertson SP. A missense mutation in ALDH18A1, encoding Delta1-pyrroline-5-carboxylate synthase (P5CS), causes an autosomal recessive neurocutaneous syndrome. Eur J Hum Genet. 2008;16(10):1176–1186. - PubMed
-
- Callewaert BL, Willaert A, Kerstjens-Frederikse WS, De Backer J, Devriendt K, Albrecht B, Ramos-Arroyo MA, Doco-Fenzy M, Hennekam RC, Pyeritz RE others. Arterial tortuosity syndrome: clinical and molecular findings in 12 newly identified families. Hum Mutat. 2008;29(1):150–158. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

