Intra- and interprotein phosphorylation between two-hybrid histidine kinases controls Myxococcus xanthus developmental progression
- PMID: 22661709
- PMCID: PMC3408162
- DOI: 10.1074/jbc.M112.387241
Intra- and interprotein phosphorylation between two-hybrid histidine kinases controls Myxococcus xanthus developmental progression
Abstract
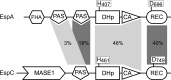
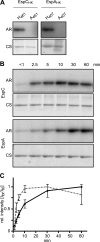
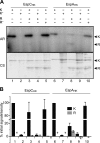
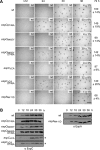
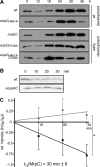
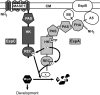
Histidine-aspartate phosphorelay signaling systems are used to couple stimuli to cellular responses. A hallmark feature is the highly modular signal transmission modules that can form both simple "two-component" systems and sophisticated multicomponent systems that integrate stimuli over time and space to generate coordinated and fine-tuned responses. The deltaproteobacterium Myxococcus xanthus contains a large repertoire of signaling proteins, many of which regulate its multicellular developmental program. Here, we assign an orphan hybrid histidine protein kinase, EspC, to the Esp signaling system that negatively regulates progression through the M. xanthus developmental program. The Esp signal system consists of the hybrid histidine protein kinase, EspA, two serine/threonine protein kinases, and a putative transport protein. We demonstrate that EspC is an essential component of this system because ΔespA, ΔespC, and ΔespA ΔespC double mutants share an identical developmental phenotype. Neither substitution of the phosphoaccepting histidine residue nor deletion of the entire catalytic ATPase domain in EspC produces an in vivo mutant developmental phenotype. In contrast, substitution of the receiver phosphoaccepting residue yields the null phenotype. Although the EspC histidine kinase can efficiently autophosphorylate in vitro, it does not act as a phosphodonor to its own receiver domain. Our in vitro and in vivo analyses suggest the phosphodonor is instead the EspA histidine kinase. We propose EspA and EspC participate in a novel hybrid histidine protein kinase signaling mechanism involving both inter- and intraprotein phosphotransfer. The output of this signaling system appears to be the combined phosphorylated state of the EspA and EspC receiver modules. This system regulates the proteolytic turnover of MrpC, an important regulator of the developmental program.
Figures


Similar articles
-
EspA, an orphan hybrid histidine protein kinase, regulates the timing of expression of key developmental proteins of Myxococcus xanthus.J Bacteriol. 2008 Jul;190(13):4416-26. doi: 10.1128/JB.00265-08. Epub 2008 Apr 4. J Bacteriol. 2008. PMID: 18390653 Free PMC article.
-
Orphan Hybrid Histidine Protein Kinase SinK Acts as a Signal Integrator To Fine-Tune Multicellular Behavior in Myxococcus xanthus.J Bacteriol. 2019 Feb 25;201(6):e00561-18. doi: 10.1128/JB.00561-18. Print 2019 Mar 15. J Bacteriol. 2019. PMID: 30617244 Free PMC article.
-
CrdS and CrdA comprise a two-component system that is cooperatively regulated by the Che3 chemosensory system in Myxococcus xanthus.mBio. 2011 Aug 2;2(4):e00110-11. doi: 10.1128/mBio.00110-11. Print 2011. mBio. 2011. PMID: 21810965 Free PMC article.
-
Dual regulation with Ser/Thr kinase cascade and a His/Asp TCS in Myxococcus xanthus.Adv Exp Med Biol. 2008;631:111-21. doi: 10.1007/978-0-387-78885-2_7. Adv Exp Med Biol. 2008. PMID: 18792684 Review.
-
Eukaryotic-like protein serine/threonine kinases in Myxococcus xanthus, a developmental bacterium exhibiting social behavior.J Cell Biochem. 1993 Jan;51(1):29-33. doi: 10.1002/jcb.240510107. J Cell Biochem. 1993. PMID: 8432741 Review.
Cited by
-
Transcription factor MrpC binds to promoter regions of hundreds of developmentally-regulated genes in Myxococcus xanthus.BMC Genomics. 2014 Dec 16;15:1123. doi: 10.1186/1471-2164-15-1123. BMC Genomics. 2014. PMID: 25515642 Free PMC article.
-
Signaling between two interacting sensor kinases promotes biofilms and colonization by a bacterial symbiont.Mol Microbiol. 2015 Apr;96(2):233-48. doi: 10.1111/mmi.12932. Epub 2015 Feb 11. Mol Microbiol. 2015. PMID: 25586643 Free PMC article.
-
Multiple communication mechanisms between sensor kinases are crucial for virulence in Pseudomonas aeruginosa.Nat Commun. 2018 Jun 7;9(1):2219. doi: 10.1038/s41467-018-04640-8. Nat Commun. 2018. PMID: 29880803 Free PMC article.
-
Mutation of self-binding sites in the promoter of the MrpC transcriptional regulator leads to asynchronous Myxococcus xanthus development.Front Microbiol. 2023 Nov 23;14:1293966. doi: 10.3389/fmicb.2023.1293966. eCollection 2023. Front Microbiol. 2023. PMID: 38075919 Free PMC article.
-
Transcriptome dynamics of the Myxococcus xanthus multicellular developmental program.Elife. 2019 Oct 14;8:e50374. doi: 10.7554/eLife.50374. Elife. 2019. PMID: 31609203 Free PMC article.
References
-
- Stock A. M., Robinson V. L., Goudreau P. N. (2000) Two-component signal transduction. Annu. Rev. Biochem. 69, 183–215 - PubMed
-
- Dutta R., Qin L., Inouye M. (1999) Histidine kinases. Diversity of domain organization. Mol. Microbiol. 34, 633–640 - PubMed
-
- Casino P., Rubio V., Marina A. (2009) Structural insight into partner specificity and phosphoryl transfer in two-component signal transduction. Cell 139, 325–336 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

