ADAMTSL4, a secreted glycoprotein widely distributed in the eye, binds fibrillin-1 microfibrils and accelerates microfibril biogenesis
- PMID: 21989719
- PMCID: PMC3292378
- DOI: 10.1167/iovs.10-5955
ADAMTSL4, a secreted glycoprotein widely distributed in the eye, binds fibrillin-1 microfibrils and accelerates microfibril biogenesis
Abstract
Purpose: ADAMTSL4 mutations cause autosomal recessive isolated ectopia lentis (IEL) and ectopia lentis et pupillae. Dominant FBN1 mutations cause IEL or syndromic ectopia lentis (Marfan syndrome and Weill-Marchesani syndrome). The authors sought to characterize recombinant ADAMTSL4 and the ocular distribution of ADAMTSL4 and to investigate whether ADAMTSL4 influences the biogenesis of fibrillin-1 microfibrils, which compose the zonule.
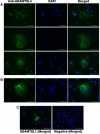
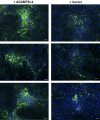
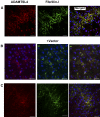
Methods: ADAMTSL4 was expressed by the transfection of HEK293F cells. Protein extracts and paraffin sections from human eyes were analyzed by Western blot analysis and by immunoperoxidase staining, respectively. Immunofluorescence was used to evaluate fibrillin-1 deposition in the ECM of fetal bovine nuchal ligament cells after culture in ADAMTSL4-conditioned medium or control medium. Confocal microscopy was performed to investigate ADAMTSL4 and fibrillin-1 colocalization in these cultures.
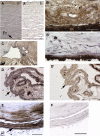
Results: Western blot analysis identified ADAMTSL4 as a glycoprotein in HEK293F cells and as a major band of 150 kDa in ocular tissues including ciliary body, sclera, cornea, and retina. Immunoperoxidase staining showed a broad ocular distribution of ADAMTSL4, associated with both cells and fibrillar ECM. When cultured in ADAMTSL4-containing medium, fetal bovine nuchal ligament cells showed accelerated fibrillin-1 deposition in ECM. ADAMTSL4 colocalized with fibrillin-1 microfibrils in the ECM of these cells.
Conclusions: ADAMTSL4 is a secreted glycoprotein that is widely distributed in the human eye. Enhanced fibrillin-1 deposition in the presence of ADAMTSL4 and colocalization of ADAMTSL4 with fibrillin-1 in the ECM of cultured fibroblasts suggest a potential role for ADAMTSL4 in the formation or maintenance of the zonule.
Figures
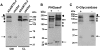
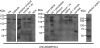

Similar articles
-
Genetic and functional linkage between ADAMTS superfamily proteins and fibrillin-1: a novel mechanism influencing microfibril assembly and function.Cell Mol Life Sci. 2011 Oct;68(19):3137-48. doi: 10.1007/s00018-011-0780-9. Epub 2011 Aug 20. Cell Mol Life Sci. 2011. PMID: 21858451 Free PMC article. Review.
-
Nonselective assembly of fibrillin 1 and fibrillin 2 in the rodent ocular zonule and in cultured cells: implications for Marfan syndrome.Invest Ophthalmol Vis Sci. 2013 Dec 23;54(13):8337-44. doi: 10.1167/iovs.13-13121. Invest Ophthalmol Vis Sci. 2013. PMID: 24265020 Free PMC article.
-
ADAMTS proteins as modulators of microfibril formation and function.Matrix Biol. 2015 Sep;47:34-43. doi: 10.1016/j.matbio.2015.05.004. Epub 2015 May 7. Matrix Biol. 2015. PMID: 25957949 Free PMC article. Review.
-
ADAMTS10 protein interacts with fibrillin-1 and promotes its deposition in extracellular matrix of cultured fibroblasts.J Biol Chem. 2011 May 13;286(19):17156-67. doi: 10.1074/jbc.M111.231571. Epub 2011 Mar 14. J Biol Chem. 2011. PMID: 21402694 Free PMC article.
-
A genotype-phenotype comparison of ADAMTSL4 and FBN1 in isolated ectopia lentis.Invest Ophthalmol Vis Sci. 2012 Jul 24;53(8):4889-96. doi: 10.1167/iovs.12-9874. Invest Ophthalmol Vis Sci. 2012. PMID: 22736615
Cited by
-
The conserved ADAMTS-like protein lonely heart mediates matrix formation and cardiac tissue integrity.PLoS Genet. 2013;9(7):e1003616. doi: 10.1371/journal.pgen.1003616. Epub 2013 Jul 11. PLoS Genet. 2013. PMID: 23874219 Free PMC article.
-
Autosomal Recessive ADAMTSL4-Related Isolated Ectopia Lentis in the Ohio Old Order Amish and Mennonite Communities.J Vitreoretin Dis. 2024 Apr 29;8(4):442-451. doi: 10.1177/24741264241249024. eCollection 2024 Jul-Aug. J Vitreoretin Dis. 2024. PMID: 39148561
-
Proteolysis of fibrillin-2 microfibrils is essential for normal skeletal development.Elife. 2022 May 3;11:e71142. doi: 10.7554/eLife.71142. Elife. 2022. PMID: 35503090 Free PMC article.
-
Review of Recurrently Mutated Genes in Craniosynostosis Supports Expansion of Diagnostic Gene Panels.Genes (Basel). 2023 Feb 28;14(3):615. doi: 10.3390/genes14030615. Genes (Basel). 2023. PMID: 36980886 Free PMC article.
-
Fell-Muir Lecture: Fibrillin microfibrils: structural tensometers of elastic tissues?Int J Exp Pathol. 2017 Aug;98(4):172-190. doi: 10.1111/iep.12239. Epub 2017 Sep 14. Int J Exp Pathol. 2017. PMID: 28905442 Free PMC article. Review.
References
-
- Cain SA, Morgan A, Sherratt MJ, Ball SG, Shuttleworth CA, Kielty CM. Proteomic analysis of fibrillin-rich microfibrils. Proteomics. 2006;6:111–122 - PubMed
-
- Hanssen E, Franc S, Garrone R. Synthesis and structural organization of zonular fibers during development and aging. Matrix Biol. 2001;20:77–85 - PubMed
-
- Wheatley HM, Traboulsi EI, Flowers BE, et al. Immunohistochemical localization of fibrillin in human ocular tissues: relevance to the Marfan syndrome. Arch Ophthalmol. 1995;113:103–109 - PubMed
-
- Kielty CM, Sherratt MJ, Marson A, Baldock C. Fibrillin microfibrils. Adv Protein Chem. 2005;70:405–436 - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases
Miscellaneous

