Absence of an orphan mitochondrial protein, c19orf12, causes a distinct clinical subtype of neurodegeneration with brain iron accumulation
- PMID: 21981780
- PMCID: PMC3188837
- DOI: 10.1016/j.ajhg.2011.09.007
Absence of an orphan mitochondrial protein, c19orf12, causes a distinct clinical subtype of neurodegeneration with brain iron accumulation
Abstract
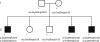
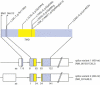
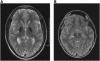
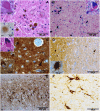
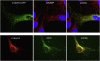
The disease classification neurodegeneration with brain iron accumulation (NBIA) comprises a clinically and genetically heterogeneous group of progressive neurodegenerative disorders characterized by brain iron deposits in the basal ganglia. For about half of the cases, the molecular basis is currently unknown. We used homozygosity mapping followed by candidate gene sequencing to identify a homozygous 11 bp deletion in the orphan gene C19orf12. Mutation screening of 23 ideopathic NBIA index cases revealed two mutated alleles in 18 of them, and one loss-of-function mutation is the most prevalent. We also identified compound heterozygous missense mutations in a case initially diagnosed with Parkinson disease at age 49. Psychiatric signs, optic atrophy, and motor axonal neuropathy were common findings. Compared to the most prevalent NBIA subtype, pantothenate kinase associated neurodegeneration (PKAN), individuals with two C19orf12 mutations were older at age of onset and the disease progressed more slowly. A polyclonal antibody against the predicted membrane spanning protein showed a mitochondrial localization. A histopathological examination in a single autopsy case detected Lewy bodies, tangles, spheroids, and tau pathology. The mitochondrial localization together with the immunohistopathological findings suggests a pathomechanistic overlap with common forms of neurodegenerative disorders.
Copyright © 2011 The American Society of Human Genetics. Published by Elsevier Inc. All rights reserved.
Figures

Similar articles
-
C19orf12 gene mutations in patients with neurodegeneration with brain iron accumulation.Parkinsonism Relat Disord. 2015 Jul;21(7):813-6. doi: 10.1016/j.parkreldis.2015.04.009. Epub 2015 Apr 23. Parkinsonism Relat Disord. 2015. PMID: 25962551
-
Analysis of the C19orf12 and WDR45 genes in patients with neurodegeneration with brain iron accumulation.J Neurol Sci. 2015 Feb 15;349(1-2):105-9. doi: 10.1016/j.jns.2014.12.036. Epub 2015 Jan 3. J Neurol Sci. 2015. PMID: 25592411
-
Neurodegeneration with brain iron accumulation.Handb Clin Neurol. 2018;147:293-305. doi: 10.1016/B978-0-444-63233-3.00019-1. Handb Clin Neurol. 2018. PMID: 29325618 Free PMC article. Review.
-
New NBIA subtype: genetic, clinical, pathologic, and radiographic features of MPAN.Neurology. 2013 Jan 15;80(3):268-75. doi: 10.1212/WNL.0b013e31827e07be. Epub 2012 Dec 26. Neurology. 2013. PMID: 23269600 Free PMC article.
-
Mitochondrial membrane protein-associated neurodegeneration (MPAN).Int Rev Neurobiol. 2013;110:73-84. doi: 10.1016/B978-0-12-410502-7.00004-1. Int Rev Neurobiol. 2013. PMID: 24209434 Review.
Cited by
-
Autosomal dominant mitochondrial membrane protein-associated neurodegeneration (MPAN).Mol Genet Genomic Med. 2019 Jul;7(7):e00736. doi: 10.1002/mgg3.736. Epub 2019 May 13. Mol Genet Genomic Med. 2019. PMID: 31087512 Free PMC article.
-
A Case of MPAN with "Eye of the Tiger Sign," Mimicking PKAN.Mov Disord Clin Pract. 2022 Jun 23;9(5):693-695. doi: 10.1002/mdc3.13493. eCollection 2022 Jul. Mov Disord Clin Pract. 2022. PMID: 35844290 Free PMC article. No abstract available.
-
An Update on the Hereditary Spastic Paraplegias: New Genes and New Disease Models.Mov Disord Clin Pract. 2015 Jun 2;2(3):213-223. doi: 10.1002/mdc3.12184. eCollection 2015 Sep. Mov Disord Clin Pract. 2015. PMID: 30838228 Free PMC article. Review.
-
The clinical spectrum of inherited diseases involved in the synthesis and remodeling of complex lipids. A tentative overview.J Inherit Metab Dis. 2015 Jan;38(1):19-40. doi: 10.1007/s10545-014-9776-6. Epub 2014 Nov 21. J Inherit Metab Dis. 2015. PMID: 25413954 Review.
-
Hereditary spastic paraplegia type 43 (SPG43) is caused by mutation in C19orf12.Hum Mutat. 2013 Oct;34(10):1357-60. doi: 10.1002/humu.22378. Epub 2013 Aug 12. Hum Mutat. 2013. PMID: 23857908 Free PMC article.
References
-
- Zhou B., Westaway S.K., Levinson B., Johnson M.A., Gitschier J., Hayflick S.J. A novel pantothenate kinase gene (PANK2) is defective in Hallervorden-Spatz syndrome. Nat. Genet. 2001;28:345–349. - PubMed
-
- Hörtnagel K., Prokisch H., Meitinger T. An isoform of hPANK2, deficient in pantothenate kinase-associated neurodegeneration, localizes to mitochondria. Hum. Mol. Genet. 2003;12:321–327. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

