Gβ5-RGS complexes are gatekeepers of hyperactivity involved in control of multiple neurotransmitter systems
- PMID: 21766168
- PMCID: PMC3260372
- DOI: 10.1007/s00213-011-2409-y
Gβ5-RGS complexes are gatekeepers of hyperactivity involved in control of multiple neurotransmitter systems
Abstract
Rationale and objectives: Our knowledge about genes involved in the control of basal motor activity that may contribute to the pathology of the hyperactivity disorders, e.g., attention deficit hyperactivity disorder (ADHD), is limited. Disruption of monoamine neurotransmitter signaling through G protein-coupled receptors (GPCR) is considered to be a major contributing factor to the etiology of the ADHD. Genetic association evidence and functional data suggest that regulators of G protein signaling proteins of the R7 family (R7 RGS) that form obligatory complexes with type 5 G protein beta subunit (Gβ5) and negatively regulate signaling downstream from monoamine GPCRs may play a role in controlling hyperactivity.
Methods: To test this hypothesis, we conducted behavioral, pharmacological, and neurochemical studies using a genetic mouse model that lacked Gβ5, a subunit essential for the expression of the entire R7 RGS family.
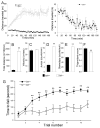
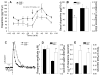
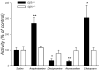
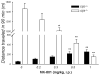
Results: Elimination of Gβ5-RGS complexes led to a striking level of hyperactivity that far exceeds activity levels previously observed in animal models. This hyperactivity was accompanied by motor learning deficits and paradoxical behavioral sensitization to a novel environment. Neurochemical studies indicated that Gβ5-RGS-deficient mice had higher sensitivity of inhibitory GPCR signaling and deficits in basal levels, release, and reuptake of dopamine. Surprisingly, pharmacological treatment with monoamine reuptake inhibitors failed to alter hyperactivity. In contrast, blockade of NMDA receptors reversed the expression of hyperactivity in Gβ5-RGS-deficient mice.
Conclusions: These findings establish that Gβ5-RGS complexes are critical regulators of monoamine-NMDA receptor signaling cross-talk and link these complexes to disorders that manifest as hyperactivity, impaired learning, and motor dysfunctions.
Figures

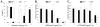
Similar articles
-
Postnatal induction and localization of R7BP, a membrane-anchoring protein for regulator of G protein signaling 7 family-Gbeta5 complexes in brain.Neuroscience. 2008 Feb 19;151(4):969-82. doi: 10.1016/j.neuroscience.2007.11.045. Epub 2007 Dec 8. Neuroscience. 2008. PMID: 18248908 Free PMC article.
-
Changes in striatal signaling induce remodeling of RGS complexes containing Gbeta5 and R7BP subunits.Mol Cell Biol. 2009 Jun;29(11):3033-44. doi: 10.1128/MCB.01449-08. Epub 2009 Mar 30. Mol Cell Biol. 2009. PMID: 19332565 Free PMC article.
-
A conserved protein interaction interface on the type 5 G protein beta subunit controls proteolytic stability and activity of R7 family regulator of G protein signaling proteins.J Biol Chem. 2010 Dec 24;285(52):41100-12. doi: 10.1074/jbc.M110.163600. Epub 2010 Oct 19. J Biol Chem. 2010. PMID: 20959458 Free PMC article.
-
Structure, function, and localization of Gβ5-RGS complexes.Prog Mol Biol Transl Sci. 2009;86:157-203. doi: 10.1016/S1877-1173(09)86006-7. Epub 2009 Oct 7. Prog Mol Biol Transl Sci. 2009. PMID: 20374716 Free PMC article. Review.
-
The R7 RGS protein family: multi-subunit regulators of neuronal G protein signaling.Cell Biochem Biophys. 2009;54(1-3):33-46. doi: 10.1007/s12013-009-9052-9. Epub 2009 Jun 12. Cell Biochem Biophys. 2009. PMID: 19521673 Free PMC article. Review.
Cited by
-
A Global Map of G Protein Signaling Regulation by RGS Proteins.Cell. 2020 Oct 15;183(2):503-521.e19. doi: 10.1016/j.cell.2020.08.052. Epub 2020 Oct 1. Cell. 2020. PMID: 33007266 Free PMC article.
-
Ablation of the GNB3 gene in mice does not affect body weight, metabolism or blood pressure, but causes bradycardia.Cell Signal. 2014 Nov;26(11):2514-20. doi: 10.1016/j.cellsig.2014.07.030. Epub 2014 Aug 2. Cell Signal. 2014. PMID: 25093805 Free PMC article.
-
A High-Throughput Time-Resolved Fluorescence Energy Transfer Assay to Screen for Modulators of RGS7/Gβ5/R7BP Complex.Assay Drug Dev Technol. 2018 Apr;16(3):150-161. doi: 10.1089/adt.2017.839. Assay Drug Dev Technol. 2018. PMID: 29658790 Free PMC article.
-
Methylomic analysis of salivary DNA in childhood ADHD identifies altered DNA methylation in VIPR2.J Child Psychol Psychiatry. 2016 Feb;57(2):152-60. doi: 10.1111/jcpp.12457. Epub 2015 Aug 25. J Child Psychol Psychiatry. 2016. PMID: 26304033 Free PMC article.
-
Inhibition of G-protein signalling in cardiac dysfunction of intellectual developmental disorder with cardiac arrhythmia (IDDCA) syndrome.J Med Genet. 2021 Dec;58(12):815-831. doi: 10.1136/jmedgenet-2020-107015. Epub 2020 Nov 10. J Med Genet. 2021. PMID: 33172956 Free PMC article.
References
-
- Anderson GR, Lujan R, Semenov A, Pravetoni M, Posokhova EN, Song JH, Uversky V, Chen CK, Wickman K, Martemyanov KA. Expression and localization of RGS9-2/G 5/R7BP complex in vivo is set by dynamic control of its constitutive degradation by cellular cysteine proteases. J Neurosci. 2007;27:14117–27. - PMC - PubMed
-
- Arnsten AF. Toward a new understanding of attention-deficit hyperactivity disorder pathophysiology: an important role for prefrontal cortex dysfunction. CNS Drugs. 2009;23(Suppl 1):33–41. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

