Loss of Bardet-Biedl syndrome protein-8 (BBS8) perturbs olfactory function, protein localization, and axon targeting
- PMID: 21646512
- PMCID: PMC3121838
- DOI: 10.1073/pnas.1016531108
Loss of Bardet-Biedl syndrome protein-8 (BBS8) perturbs olfactory function, protein localization, and axon targeting
Abstract
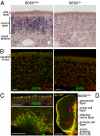
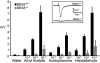
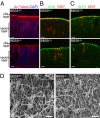
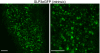
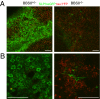
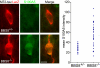
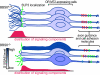
Bardet-Biedl syndrome (BBS) is a pleiotropic, heterogeneous human disease whose etiology lies primarily in dysfunctional basal bodies and/or cilia. Both BBS patients and several BBS mouse models exhibit impaired olfactory function. To explore the nature of olfactory defects in BBS, a genetic ablation of the mouse Bbs8 gene that incorporates a fluorescent reporter protein was created. The endogenous BBS8 protein and reporter are particularly abundant in olfactory sensory neurons (OSNs), and specific BBS8 antibodies reveal staining in the dendritic knob in a shell-like structure that surrounds the basal bodies. Bbs8-null mice have reduced olfactory responses to a number of odorants, and immunohistochemical analyses reveal a near-complete loss of cilia from OSNs and mislocalization of proteins normally enriched in cilia. To visualize altered protein localization in OSNs, we generated a SLP3(eGFP) knock-in mouse and imaged the apical epithelium, including dendritic knobs and proximal cilia, in ex vivo tissue preparations. Additionally, protein reagents that reflect the characteristic neuronal activity of each OSN revealed altered activity in Bbs8-null cells. In addition to previously known defects at the ciliary border, we also observed aberrant targeting of OSN axons to the olfactory bulb; axons expressing the same receptor display reduced fasciculation and project to multiple targets in the olfactory bulb. We suggest that loss of BBS8 leads to a dramatic and variable reduction in cilia, the essential signaling platform for olfaction, which alters the uniformity of responses in populations of OSNs expressing the same receptor, thereby contributing to the observed axon-targeting defects.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
BBS4 is required for intraflagellar transport coordination and basal body number in mammalian olfactory cilia.J Cell Sci. 2019 Feb 15;132(5):jcs222331. doi: 10.1242/jcs.222331. J Cell Sci. 2019. PMID: 30665891 Free PMC article.
-
Gene Therapeutic Reversal of Peripheral Olfactory Impairment in Bardet-Biedl Syndrome.Mol Ther. 2017 Apr 5;25(4):904-916. doi: 10.1016/j.ymthe.2017.02.006. Epub 2017 Feb 22. Mol Ther. 2017. PMID: 28237838 Free PMC article.
-
Loss of BBS proteins causes anosmia in humans and defects in olfactory cilia structure and function in the mouse.Nat Genet. 2004 Sep;36(9):994-8. doi: 10.1038/ng1418. Epub 2004 Aug 22. Nat Genet. 2004. PMID: 15322545
-
Establishing a connection between cilia and Bardet-Biedl Syndrome.Trends Mol Med. 2004 Mar;10(3):106-9. doi: 10.1016/j.molmed.2004.01.003. Trends Mol Med. 2004. PMID: 15106604 Review.
-
Maturation of the Olfactory Sensory Neuron and Its Cilia.Chem Senses. 2020 Dec 5;45(9):805-822. doi: 10.1093/chemse/bjaa070. Chem Senses. 2020. PMID: 33075817 Free PMC article. Review.
Cited by
-
Correction of cilia structure and function alleviates multi-organ pathology in Bardet-Biedl syndrome mice.Hum Mol Genet. 2020 Aug 29;29(15):2508-2522. doi: 10.1093/hmg/ddaa138. Hum Mol Genet. 2020. PMID: 32620959 Free PMC article.
-
Insights into the molecular triggers of parosmia based on gas chromatography olfactometry.Commun Med (Lond). 2022 May 24;2:58. doi: 10.1038/s43856-022-00112-9. eCollection 2022. Commun Med (Lond). 2022. PMID: 35647609 Free PMC article.
-
Gene therapy rescues olfactory perception in a clinically relevant ciliopathy model of Bardet-Biedl syndrome.FASEB J. 2021 Sep;35(9):e21766. doi: 10.1096/fj.202100627R. FASEB J. 2021. PMID: 34383976 Free PMC article.
-
Serotonin 5-HT6 receptors affect cognition in a mouse model of Alzheimer's disease by regulating cilia function.Alzheimers Res Ther. 2017 Sep 20;9(1):76. doi: 10.1186/s13195-017-0304-4. Alzheimers Res Ther. 2017. PMID: 28931427 Free PMC article.
-
A Role for STOML3 in Olfactory Sensory Transduction.eNeuro. 2021 Mar 12;8(2):ENEURO.0565-20.2021. doi: 10.1523/ENEURO.0565-20.2021. Print 2021 Mar-Apr. eNeuro. 2021. PMID: 33637538 Free PMC article.
References
-
- Ansley SJ, et al. Basal body dysfunction is a likely cause of pleiotropic Bardet-Biedl syndrome. Nature. 2003;425:628–633. - PubMed
-
- Mokrzan EM, Lewis JS, Mykytyn K. Differences in renal tubule primary cilia length in a mouse model of Bardet-Biedl syndrome. Nephron Exp Nephrol. 2007;106:e88–e96. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

