Severe congenital neutropenia resulting from G6PC3 deficiency with increased neutrophil CXCR4 expression and myelokathexis
- PMID: 20616219
- PMCID: PMC2974587
- DOI: 10.1182/blood-2010-01-265942
Severe congenital neutropenia resulting from G6PC3 deficiency with increased neutrophil CXCR4 expression and myelokathexis
Abstract
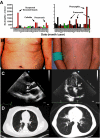
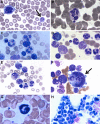
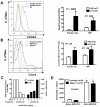
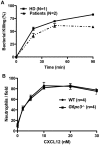
Mutations in more than 15 genes are now known to cause severe congenital neutropenia (SCN); however, the pathologic mechanisms of most genetic defects are not fully defined. Deficiency of G6PC3, a glucose-6-phosphatase, causes a rare multisystem syndrome with SCN first described in 2009. We identified a family with 2 children with homozygous G6PC3 G260R mutations, a loss of enzymatic function, and typical syndrome features with the exception that their bone marrow biopsy pathology revealed abundant neutrophils consistent with myelokathexis. This pathologic finding is a hallmark of another type of SCN, WHIM syndrome, which is caused by gain-of-function mutations in CXCR4, a chemokine receptor and known neutrophil bone marrow retention factor. We found markedly increased CXCR4 expression on neutrophils from both our G6PC3-deficient patients and G6pc3(-/-) mice. In both patients, granulocyte colony-stimulating factor treatment normalized CXCR4 expression and neutrophil counts. In G6pc3(-/-) mice, the specific CXCR4 antagonist AMD3100 rapidly reversed neutropenia. Thus, myelokathexis associated with abnormally high neutrophil CXCR4 expression may contribute to neutropenia in G6PC3 deficiency and responds well to granulocyte colony-stimulating factor.
Figures
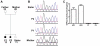

Similar articles
-
Lack of glucose recycling between endoplasmic reticulum and cytoplasm underlies cellular dysfunction in glucose-6-phosphatase-beta-deficient neutrophils in a congenital neutropenia syndrome.Blood. 2010 Oct 14;116(15):2783-92. doi: 10.1182/blood-2009-12-258491. Epub 2010 May 24. Blood. 2010. PMID: 20498302 Free PMC article.
-
A clinical and molecular review of ubiquitous glucose-6-phosphatase deficiency caused by G6PC3 mutations.Orphanet J Rare Dis. 2013 Jun 13;8:84. doi: 10.1186/1750-1172-8-84. Orphanet J Rare Dis. 2013. PMID: 23758768 Free PMC article. Review.
-
A syndrome with congenital neutropenia and mutations in G6PC3.N Engl J Med. 2009 Jan 1;360(1):32-43. doi: 10.1056/NEJMoa0805051. N Engl J Med. 2009. PMID: 19118303 Free PMC article.
-
A novel G6PC3 gene mutation in a patient with severe congenital neutropenia.J Pediatr Hematol Oncol. 2013 Mar;35(2):e81-3. doi: 10.1097/MPH.0b013e3182679000. J Pediatr Hematol Oncol. 2013. PMID: 23018568
-
Novel G6PC3 Mutations in Patients with Congenital Neutropenia: Case Reports and Review of the Literature.Endocr Metab Immune Disord Drug Targets. 2021;21(9):1660-1668. doi: 10.2174/1871530321666210616110631. Endocr Metab Immune Disord Drug Targets. 2021. PMID: 34137364 Review.
Cited by
-
CXCR4 antagonist AMD3100 redistributes leukocytes from primary immune organs to secondary immune organs, lung, and blood in mice.Eur J Immunol. 2015 Jun;45(6):1855-67. doi: 10.1002/eji.201445245. Epub 2015 Apr 24. Eur J Immunol. 2015. PMID: 25801950 Free PMC article.
-
Intestinal Immune Imbalance is an Alarm in the Development of IBD.Mediators Inflamm. 2023 Jul 31;2023:1073984. doi: 10.1155/2023/1073984. eCollection 2023. Mediators Inflamm. 2023. PMID: 37554552 Free PMC article. Review.
-
Understanding neutropenia secondary to intrinsic or iatrogenic immune dysregulation.Hematology Am Soc Hematol Educ Program. 2021 Dec 10;2021(1):504-513. doi: 10.1182/hematology.2021000285. Hematology Am Soc Hematol Educ Program. 2021. PMID: 34889406 Free PMC article.
-
A novel homozygous mutation in G6PC3 presenting as cyclic neutropenia and severe congenital neutropenia in the same family.J Clin Immunol. 2013 Nov;33(8):1403-6. doi: 10.1007/s10875-013-9945-7. Epub 2013 Oct 9. J Clin Immunol. 2013. PMID: 24105461
-
Type I glycogen storage diseases: disorders of the glucose-6-phosphatase/glucose-6-phosphate transporter complexes.J Inherit Metab Dis. 2015 May;38(3):511-9. doi: 10.1007/s10545-014-9772-x. Epub 2014 Oct 7. J Inherit Metab Dis. 2015. PMID: 25288127 Review.
References
-
- Bohn G, Welte K, Klein C. Severe congenital neutropenia: new genes explain an old disease. Curr Opin Rheumatol. 2007;19(6):644–650. - PubMed
-
- Kostmann R. Infantile genetic agranulocytosis; agranulocytosis infantilis hereditaria. Acta Paediatr Suppl. 1956;45(suppl 105):1–78. - PubMed
-
- Ancliff PJ, Blundell MP, Cory GO, et al. Two novel activating mutations in the Wiskott-Aldrich syndrome protein result in congenital neutropenia. Blood. 2006;108(7):2182–2189. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

