Myxobacteria, polarity, and multicellular morphogenesis
- PMID: 20610548
- PMCID: PMC2908774
- DOI: 10.1101/cshperspect.a000380
Myxobacteria, polarity, and multicellular morphogenesis
Abstract
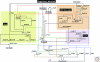
Myxobacteria are renowned for the ability to sporulate within fruiting bodies whose shapes are species-specific. The capacity to build those multicellular structures arises from the ability of M. xanthus to organize high cell-density swarms, in which the cells tend to be aligned with each other while constantly in motion. The intrinsic polarity of rod-shaped cells lays the foundation, and each cell uses two polar engines for gliding on surfaces. It sprouts retractile type IV pili from the leading cell pole and secretes capsular polysaccharide through nozzles from the trailing pole. Regularly periodic reversal of the gliding direction was found to be required for swarming. Those reversals are generated by a G-protein switch which is driven by a sharply tuned oscillator. Starvation induces fruiting body development, and systematic reductions in the reversal frequency are necessary for the cells to aggregate rather than continue to swarm. Developmental gene expression is regulated by a network that is connected to the suppression of reversals.
Figures
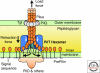


Similar articles
-
Reversing cell polarity: evidence and hypothesis.Curr Opin Microbiol. 2005 Apr;8(2):216-21. doi: 10.1016/j.mib.2005.02.002. Curr Opin Microbiol. 2005. PMID: 15802255 Review.
-
Coupling cell movement to multicellular development in myxobacteria.Nat Rev Microbiol. 2003 Oct;1(1):45-54. doi: 10.1038/nrmicro733. Nat Rev Microbiol. 2003. PMID: 15040179 Review.
-
Myxococcus-from single-cell polarity to complex multicellular patterns.Annu Rev Genet. 2008;42:109-30. doi: 10.1146/annurev.genet.42.110807.091615. Annu Rev Genet. 2008. PMID: 18605899 Review.
-
Myxococcus xanthus swarms are driven by growth and regulated by a pacemaker.J Bacteriol. 2011 Nov;193(21):5898-904. doi: 10.1128/JB.00168-11. Epub 2011 Aug 19. J Bacteriol. 2011. PMID: 21856842 Free PMC article.
-
Dynamics of fruiting body morphogenesis.J Bacteriol. 2004 Feb;186(4):919-27. doi: 10.1128/JB.186.4.919-927.2004. J Bacteriol. 2004. PMID: 14761986 Free PMC article.
Cited by
-
Ultrasensitive Response of Developing Myxococcus xanthus to the Addition of Nutrient Medium Correlates with the Level of MrpC.J Bacteriol. 2018 Oct 23;200(22):e00456-18. doi: 10.1128/JB.00456-18. Print 2018 Nov 15. J Bacteriol. 2018. PMID: 30181127 Free PMC article.
-
Heterologous Biosynthesis of Myxobacterial Antibiotic Miuraenamide A.Molecules. 2023 Mar 20;28(6):2815. doi: 10.3390/molecules28062815. Molecules. 2023. PMID: 36985787 Free PMC article.
-
Uncovering the mystery of gliding motility in the myxobacteria.Annu Rev Genet. 2011;45:21-39. doi: 10.1146/annurev-genet-110410-132547. Epub 2011 Sep 9. Annu Rev Genet. 2011. PMID: 21910630 Free PMC article. Review.
-
Profiling Myxococcus xanthus Swarming Phenotypes through Mutation and Environmental Variation.J Bacteriol. 2021 Nov 5;203(23):e0030621. doi: 10.1128/JB.00306-21. Epub 2021 Sep 20. J Bacteriol. 2021. PMID: 34543101 Free PMC article.
-
Genomic Analysis of the Rare Slightly Halophilic Myxobacterium "Paraliomyxa miuraensis" SMH-27-4, the Producer of the Antibiotic Miuraenamide A.Microorganisms. 2023 Feb 1;11(2):371. doi: 10.3390/microorganisms11020371. Microorganisms. 2023. PMID: 36838335 Free PMC article.
References
-
- Acuna G, Shi W, Trudeau K, Zusman D 1995. The cheA and cheY domains of Myxococcus xanthus FrzE function independently in vitro as an autokinase and a phosphate acceptor, respectively. FEBS Letts 358: 31–33 - PubMed
-
- Astling DP, Lee JY, Zusman DR 2006. Differential effects of chemoreceptor methylation-domain mutations on swarming and development in the social bacterium Myxococcus xanthus. Mol Microbiol 59: 45–55 - PubMed
-
- Bitter W, Koster M, Latijnhouwers M, de Cock H, Tommassen J 1998. Formation of oligomeric rings by XcpQ and PilQ, which are involved in protein transport across the outer membrane of Pseudomonas aeruginosa. Mol Microbiol 27: 209–219 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
