Galpha(i1) and Galpha(i3) are required for epidermal growth factor-mediated activation of the Akt-mTORC1 pathway
- PMID: 19401591
- PMCID: PMC4138699
- DOI: 10.1126/scisignal.2000118
Galpha(i1) and Galpha(i3) are required for epidermal growth factor-mediated activation of the Akt-mTORC1 pathway
Erratum in
- Sci Signal. 2010;3(121):er2
Abstract
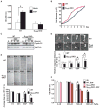
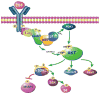
The precise mechanism whereby epidermal growth factor (EGF) activates the serine-threonine kinase Akt and the mammalian target of rapamycin (mTOR) complex 1 (mTORC1) remains elusive. Here, we report that the alpha subunits of the heterotrimeric guanine nucleotide-binding proteins (G proteins) Galpha(i1) and Galpha(i3) are critical for this activation process. Both Galpha(i1) and Galpha(i3) formed complexes with growth factor receptor binding 2 (Grb2)-associated binding protein 1 (Gab1) and the EGF receptor (EGFR) and were required for the phosphorylation of Gab1 and its subsequent interaction with the p85 subunit of phosphatidylinositol 3-kinase in response to EGF. Loss of Galpha(i1) and Galpha(i3) severely impaired the activation of Akt and of p70 S6 kinase and 4E-BP1, downstream targets of mTORC1, in response to EGF, heparin-binding EGF-like growth factor, and transforming growth factor alpha, but not insulin, insulin-like growth factor, or platelet-derived growth factor. In addition, ablation of Galpha(i1) and Galpha(i3) largely inhibited EGF-induced cell growth, migration, and survival and the accumulation of cyclin D1. Overall, this study suggests that Galpha(i1) and Galpha(i3) lie downstream of EGFR, but upstream of Gab1-mediated activation of Akt and mTORC1, thus revealing a role for Galpha(i) proteins in mediating EGFR signaling.
Figures





Similar articles
-
G(i)α proteins exhibit functional differences in the activation of ERK1/2, Akt and mTORC1 by growth factors in normal and breast cancer cells.Cell Commun Signal. 2014 Feb 13;12:10. doi: 10.1186/1478-811X-12-10. Cell Commun Signal. 2014. PMID: 24521094 Free PMC article.
-
Requirement of Gαi1/3-Gab1 signaling complex for keratinocyte growth factor-induced PI3K-AKT-mTORC1 activation.J Invest Dermatol. 2015 Jan;135(1):181-191. doi: 10.1038/jid.2014.326. Epub 2014 Jul 31. J Invest Dermatol. 2015. PMID: 25078664
-
Pertussis toxin-sensitive Gi/o proteins are involved in nerve growth factor-induced pro-survival Akt signaling cascade in PC12 cells.Cell Signal. 2005 Jul;17(7):881-90. doi: 10.1016/j.cellsig.2004.11.008. Epub 2004 Dec 15. Cell Signal. 2005. PMID: 15763430
-
Multifaceted role of mTOR (mammalian target of rapamycin) signaling pathway in human health and disease.Signal Transduct Target Ther. 2023 Oct 2;8(1):375. doi: 10.1038/s41392-023-01608-z. Signal Transduct Target Ther. 2023. PMID: 37779156 Free PMC article. Review.
-
Signaling specificity in the Akt pathway in biology and disease.Adv Biol Regul. 2014 May;55:28-38. doi: 10.1016/j.jbior.2014.04.001. Epub 2014 Apr 19. Adv Biol Regul. 2014. PMID: 24794538 Free PMC article. Review.
Cited by
-
Identification of Gαi3 as a novel molecular therapeutic target of cervical cancer.Int J Biol Sci. 2022 Sep 6;18(15):5667-5680. doi: 10.7150/ijbs.77126. eCollection 2022. Int J Biol Sci. 2022. PMID: 36263185 Free PMC article.
-
Signal protein-derived peptides as functional probes and regulators of intracellular signaling.J Amino Acids. 2011;2011:656051. doi: 10.4061/2011/656051. Epub 2011 Aug 23. J Amino Acids. 2011. PMID: 22312467 Free PMC article.
-
Overexpressed Gαi1 exerts pro-tumorigenic activity in nasopharyngeal carcinoma.Cell Death Dis. 2023 Dec 4;14(12):792. doi: 10.1038/s41419-023-06308-8. Cell Death Dis. 2023. PMID: 38049415 Free PMC article.
-
RIPK1 regulates the survival of human melanocytes upon endoplasmic reticulum stress.Exp Ther Med. 2020 May;19(5):3239-3246. doi: 10.3892/etm.2020.8575. Epub 2020 Mar 6. Exp Ther Med. 2020. PMID: 32266019 Free PMC article.
-
G(i)α proteins exhibit functional differences in the activation of ERK1/2, Akt and mTORC1 by growth factors in normal and breast cancer cells.Cell Commun Signal. 2014 Feb 13;12:10. doi: 10.1186/1478-811X-12-10. Cell Commun Signal. 2014. PMID: 24521094 Free PMC article.
References
-
- Rozengurt E. Mitogenic signaling pathways induced by G protein-coupled receptors. J Cell Physiol. 2007;213:589–602. - PubMed
-
- Steelman LS, Abrams SL, Whelan J, Bertrand FE, Ludwig DE, Bäsecke J, Libra M, Stivala F, Milella M, Tafuri A, Lunghi P, Bonati A, Martelli AM, McCubrey JA. Contributions of the Raf/MEK/ERK, PI3K/PTEN/Akt/mTOR and Jak/STAT pathways to leukemia. Leukemia. 2008;22:686–707. - PubMed
-
- Sabatini DM. mTOR and cancer: Insights into a complex relationship. Nat Rev Cancer. 2006;6:729–734. - PubMed
-
- Greer EL, Brunet A. FOXO transcription factors in ageing and cancer. Acta Physiol (Oxf) 2008;192:19–28. - PubMed
-
- Lam EW, Francis RE, Petkovic M. FOXO transcription factors: Key regulators of cell fate. Biochem Soc Trans. 2006;34:722–726. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials
Miscellaneous

