Coexpression and heteromerization of two neuronal K-Cl cotransporter isoforms in neonatal brain
- PMID: 19307176
- PMCID: PMC2679471
- DOI: 10.1074/jbc.M807366200
Coexpression and heteromerization of two neuronal K-Cl cotransporter isoforms in neonatal brain
Abstract
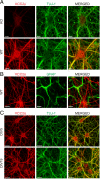
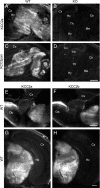
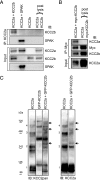
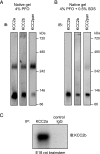
The neuron-specific K-Cl cotransporter KCC2 maintains the low intracellular chloride concentration required for the fast hyperpolarizing actions of inhibitory neurotransmitters. The KCC2 gene codes for two isoforms, KCC2a and KCC2b, which differ in their N termini. The relative expression and cellular distribution of the two KCC2 protein isoforms are unknown. Here, we characterize an antibody against the KCC2a isoform and show that a previously described antibody against KCC2 is specific for the KCC2b isoform (Hubner, C. A., Stein, V., Hermans-Borgmeyer, I., Meyer, T., Ballanyi, K., and Jentsch, T. J. (2001) Neuron 30, 515-524). Immunostaining of dissociated hippocampal cultures confirms that both KCC2 isoforms are neuron-specific. Immunoblot analysis indicates that KCC2b is the major KCC2 isoform in the adult brain, whereas in the neonatal mouse central nervous system, half of total KCC2 protein is KCC2a. At this stage, the two KCC2 isoforms are largely colocalized and show similar patterns of distribution in the brain. When coexpressed in HEK293 cells, KCC2a and KCC2b proteins form heteromeric complexes. Moreover, the two isoforms can be coimmunoprecipitated from the neonatal brain, suggesting the presence of endogenous KCC2a-KCC2b heteromers. Consistent with this, native gel analysis shows that a substantial part of endogenous KCC2 isoforms in the neonatal brain constitute dimers.
Figures

Similar articles
-
Distribution of neuronal KCC2a and KCC2b isoforms in mouse CNS.J Comp Neurol. 2014 Jun 1;522(8):1897-914. doi: 10.1002/cne.23510. J Comp Neurol. 2014. PMID: 24639001
-
A novel N-terminal isoform of the neuron-specific K-Cl cotransporter KCC2.J Biol Chem. 2007 Oct 19;282(42):30570-6. doi: 10.1074/jbc.M705095200. Epub 2007 Aug 22. J Biol Chem. 2007. PMID: 17715129
-
Implications of the N-terminal heterogeneity for the neuronal K-Cl cotransporter KCC2 function.Brain Res. 2017 Nov 15;1675:87-101. doi: 10.1016/j.brainres.2017.08.034. Epub 2017 Sep 6. Brain Res. 2017. PMID: 28888841
-
Two developmental switches in GABAergic signalling: the K+-Cl- cotransporter KCC2 and carbonic anhydrase CAVII.J Physiol. 2005 Jan 1;562(Pt 1):27-36. doi: 10.1113/jphysiol.2004.077495. Epub 2004 Nov 4. J Physiol. 2005. PMID: 15528236 Free PMC article. Review.
-
Role of NKCC1 and KCC2 in Epilepsy: From Expression to Function.Front Neurol. 2020 Jan 17;10:1407. doi: 10.3389/fneur.2019.01407. eCollection 2019. Front Neurol. 2020. PMID: 32010056 Free PMC article. Review.
Cited by
-
Contribution of the potassium-chloride cotransporter KCC2 to the strength of inhibition in the neonatal rodent spinal cord in vitro.J Neurosci. 2015 Apr 1;35(13):5307-16. doi: 10.1523/JNEUROSCI.1674-14.2015. J Neurosci. 2015. PMID: 25834055 Free PMC article.
-
Refuting the challenges of the developmental shift of polarity of GABA actions: GABA more exciting than ever!Front Cell Neurosci. 2012 Aug 28;6:35. doi: 10.3389/fncel.2012.00035. eCollection 2012. Front Cell Neurosci. 2012. PMID: 22973192 Free PMC article.
-
Chloride Homeostasis in Developing Motoneurons.Adv Neurobiol. 2022;28:45-61. doi: 10.1007/978-3-031-07167-6_2. Adv Neurobiol. 2022. PMID: 36066820
-
Rett and Rett-related disorders: Common mechanisms for shared symptoms?Exp Biol Med (Maywood). 2023 Nov;248(22):2095-2108. doi: 10.1177/15353702231209419. Epub 2023 Dec 6. Exp Biol Med (Maywood). 2023. PMID: 38057990 Free PMC article. Review.
-
Reciprocal Regulation of KCC2 Trafficking and Synaptic Activity.Front Cell Neurosci. 2019 Feb 20;13:48. doi: 10.3389/fncel.2019.00048. eCollection 2019. Front Cell Neurosci. 2019. PMID: 30842727 Free PMC article. Review.
References
-
- Blaesse, P., Airaksinen, M. S., Rivera, C., and Kaila, K. (2009) Neuron 61 820-838 - PubMed
-
- Payne, J. A., Stevenson, T. J., and Donaldson, L. F. (1996) J. Biol. Chem. 271 16245-16252 - PubMed
-
- Gamba, G. (2005) Physiol. Rev. 85 423-493 - PubMed
-
- Casula, S., Shmukler, B. E., Wilhelm, S., Stuart-Tilley, A. K., Su, W., Chernova, M. N., Brugnara, C., and Alper, S. L. (2001) J. Biol. Chem. 276 41870-41878 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

