Mutations in LAMB2 causing a severe form of synaptic congenital myasthenic syndrome
- PMID: 19251977
- PMCID: PMC2643050
- DOI: 10.1136/jmg.2008.063693
Mutations in LAMB2 causing a severe form of synaptic congenital myasthenic syndrome
Abstract
Background: We describe a severe form of congenital myasthenic syndrome (CMS) associated with congenital nephrosis and ocular malformations caused by two truncating mutations in the gene encoding the laminin beta2 subunit (LAMB2).
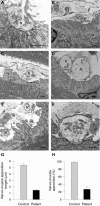
Methods and results: Mutational analysis in the affected patient, who has a history of a serious untoward reaction to treatment with acetylcholinesterase inhibition, revealed two frame-shifting heteroallelic mutations, a maternally inherited 1478delG and a paternally inherited 4804delC. An anconeus muscle biopsy demonstrated a profound distortion of the architecture and function of the neuromuscular junction, which was strikingly similar to that seen in mice lacking laminin beta2 subunit. The findings included: pronounced reduction of the axon terminal size with encasement of the nerve endings by Schwann cells, severe widening of the primary synaptic cleft and invasion of the synaptic space by the processes of Schwann cells, and moderate simplification of postsynaptic folds and intact expression of the endplate acetylcholinesterase. The endplate potential quantal content was notably reduced, while the frequencies and amplitudes of miniature endplate potentials were only moderately diminished and the decay phases of miniature endplate potentials were normal. Western blot analysis of muscle and kidney tissue and immunohistochemistry of kidney tissue showed no laminin beta2 expression.
Conclusion: This case, which represents a new type of synaptic CMS, exemplifies the wide variability of phenotypes associated with LAMB2 mutations and underscores the fundamental role that laminin beta2 plays in the development of the human neuromuscular junction.
Conflict of interest statement
Figures

Similar articles
-
LG2 agrin mutation causing severe congenital myasthenic syndrome mimics functional characteristics of non-neural (z-) agrin.Hum Genet. 2012 Jul;131(7):1123-35. doi: 10.1007/s00439-011-1132-4. Epub 2011 Dec 29. Hum Genet. 2012. PMID: 22205389 Free PMC article.
-
Synaptic basal lamina-associated congenital myasthenic syndromes.Ann N Y Acad Sci. 2012 Dec;1275:36-48. doi: 10.1111/j.1749-6632.2012.06807.x. Ann N Y Acad Sci. 2012. PMID: 23278576 Review.
-
Presynaptic congenital myasthenic syndrome with a homozygous sequence variant in LAMA5 combines myopia, facial tics, and failure of neuromuscular transmission.Am J Med Genet A. 2017 Aug;173(8):2240-2245. doi: 10.1002/ajmg.a.38291. Epub 2017 May 25. Am J Med Genet A. 2017. PMID: 28544784 Free PMC article.
-
A presynaptic congenital myasthenic syndrome attributed to a homozygous sequence variant in LAMA5.Ann N Y Acad Sci. 2018 Feb;1413(1):119-125. doi: 10.1111/nyas.13585. Epub 2018 Jan 28. Ann N Y Acad Sci. 2018. PMID: 29377152 Free PMC article. Review.
-
Mutations in MUSK causing congenital myasthenic syndrome impair MuSK-Dok-7 interaction.Hum Mol Genet. 2010 Jun 15;19(12):2370-9. doi: 10.1093/hmg/ddq110. Epub 2010 Apr 6. Hum Mol Genet. 2010. PMID: 20371544 Free PMC article.
Cited by
-
LAMB2 novel variant c.2885-9 C>A affects RNA splicing in a minigene assay.Mol Genet Genomic Med. 2021 Jul;9(7):e1704. doi: 10.1002/mgg3.1704. Epub 2021 May 13. Mol Genet Genomic Med. 2021. PMID: 33982833 Free PMC article.
-
Role of extracellular matrix proteins and their receptors in the development of the vertebrate neuromuscular junction.Dev Neurobiol. 2011 Nov;71(11):982-1005. doi: 10.1002/dneu.20953. Dev Neurobiol. 2011. PMID: 21766463 Free PMC article. Review.
-
Congenital Myasthenic Syndromes in 2018.Curr Neurol Neurosci Rep. 2018 Jun 12;18(8):46. doi: 10.1007/s11910-018-0852-4. Curr Neurol Neurosci Rep. 2018. PMID: 29892917 Review.
-
Clinical relevance of terminal Schwann cells: An overlooked component of the neuromuscular junction.J Neurosci Res. 2018 Jul;96(7):1125-1135. doi: 10.1002/jnr.24231. Epub 2018 Mar 13. J Neurosci Res. 2018. PMID: 29536564 Free PMC article. Review.
-
Basement Membrane Defects in Genetic Kidney Diseases.Front Pediatr. 2018 Jan 29;6:11. doi: 10.3389/fped.2018.00011. eCollection 2018. Front Pediatr. 2018. PMID: 29435440 Free PMC article. Review.
References
-
- Engel AG, Sine SM. Current understanding of congenital myasthenic syndromes. Curr Opin Pharmacol 2005;5:308–21 - PubMed
-
- Hantai D, Richard P, Koenig J, Eymard B. Congenital myasthenic syndromes. Curr Opin Neurol 2004;17:539–51 - PubMed
-
- Hoffmann K, Muller JS, Stricker S, Megarbane A, Rajab A, Lindner TH, Cohen M, Chouery E, Adaimy L, Ghanem I, Delaque V, Boltshauser E, Talim B, Horvath R, Robinson PN, Lochmüller H, Hübner C, Mundlos S. Escobar syndrome is a prenatal myasthenia caused by disruption of the acetylcholine receptor fetal gamma subunit. Am J Hum Genet 2006;79:303–12 - PMC - PubMed
-
- Chevessier F, Faraut B, Ravel-Chapuis A, Richard P, Gaudon K, Bauché S, Prioleau C, Herbst R, Goillot E, Ioos C, Azulay JP, Attarian S, Leroy JP, Fournier E, Legay C, Schaeffer L, Koenig J, Fardeau M, Eymard B, Pouget J, Hantai D. MUSK, a new target for mutations causing congenital myasthenic syndrome. Hum Mol Genet 2004;13:3229–40 - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
