Adverse events with bismuth salts for Helicobacter pylori eradication: systematic review and meta-analysis
- PMID: 19109870
- PMCID: PMC2778120
- DOI: 10.3748/wjg.14.7361
Adverse events with bismuth salts for Helicobacter pylori eradication: systematic review and meta-analysis
Abstract
Aim: To assess the safety of bismuth used in Helicobacter pylori (H pylori) eradication therapy regimens.
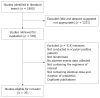
Methods: We conducted a systematic review and meta-analysis. MEDLINE and EMBASE were searched (up to October 2007) to identify randomised controlled trials comparing bismuth with placebo or no treatment, or bismuth salts in combination with antibiotics as part of eradication therapy with the same dose and duration of antibiotics alone or, in combination, with acid suppression. Total numbers of adverse events were recorded. Data were pooled and expressed as relative risks with 95% confidence intervals (CI).
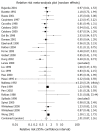
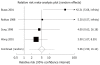
Results: We identified 35 randomised controlled trials containing 4763 patients. There were no serious adverse events occurring with bismuth therapy. There was no statistically significant difference detected in total adverse events with bismuth [relative risk (RR) = 1.01; 95% CI: 0.87-1.16], specific individual adverse events, with the exception of dark stools (RR = 5.06; 95% CI: 1.59-16.12), or adverse events leading to withdrawal of therapy (RR = 0.86; 95% CI: 0.54-1.37).
Conclusion: Bismuth for the treatment of H pylori is safe and well-tolerated. The only adverse event occurring significantly more commonly was dark stools.
Figures
Similar articles
-
Optimum duration of regimens for Helicobacter pylori eradication.Cochrane Database Syst Rev. 2013 Dec 11;2013(12):CD008337. doi: 10.1002/14651858.CD008337.pub2. Cochrane Database Syst Rev. 2013. PMID: 24338763 Free PMC article. Review.
-
Review article: bismuth-based therapy for Helicobacter pylori eradication in children.Aliment Pharmacol Ther. 2012 May;35(9):1010-26. doi: 10.1111/j.1365-2036.2012.05055.x. Epub 2012 Mar 8. Aliment Pharmacol Ther. 2012. PMID: 22404517 Review.
-
Bismuth supplements as the first-line regimen for Helicobacter pylori eradication therapy: Systemic review and meta-analysis.Helicobacter. 2019 Apr;24(2):e12565. doi: 10.1111/hel.12565. Epub 2019 Jan 30. Helicobacter. 2019. PMID: 30698318
-
Helicobacter pylori eradication: proton pump inhibitor vs. ranitidine bismuth citrate plus two antibiotics for 1 week-a meta-analysis of efficacy.Aliment Pharmacol Ther. 2000 Sep;14(9):1141-50. doi: 10.1046/j.1365-2036.2000.00809.x. Aliment Pharmacol Ther. 2000. PMID: 10971230
-
Pharmacological regimens for eradication of Helicobacter pylori: an overview of systematic reviews and network meta-analysis.BMC Gastroenterol. 2016 Jul 26;16(1):80. doi: 10.1186/s12876-016-0491-7. BMC Gastroenterol. 2016. PMID: 27460211 Free PMC article. Review.
Cited by
-
Pharmacokinetic and Safety Study of Bismuth Potassium Citrate Formulations in Healthy Subjects.Drugs R D. 2024 Mar;24(1):81-87. doi: 10.1007/s40268-024-00455-9. Epub 2024 Feb 12. Drugs R D. 2024. PMID: 38345697 Free PMC article.
-
Crystal structure, antibacterial and cytotoxic activities of a new complex of bismuth(III) with sulfapyridine.Molecules. 2013 Jan 24;18(2):1464-76. doi: 10.3390/molecules18021464. Molecules. 2013. PMID: 23348999 Free PMC article.
-
Review of current diagnostic methods and advances in Helicobacter pylori diagnostics in the era of next generation sequencing.World J Gastroenterol. 2019 Aug 28;25(32):4629-4660. doi: 10.3748/wjg.v25.i32.4629. World J Gastroenterol. 2019. PMID: 31528091 Free PMC article. Review.
-
Quadruple therapy with moxifloxacin and bismuth for first-line treatment of Helicobacter pylori.World J Gastroenterol. 2012 Aug 28;18(32):4386-90. doi: 10.3748/wjg.v18.i32.4386. World J Gastroenterol. 2012. PMID: 22969203 Free PMC article. Clinical Trial.
-
Failure or success of electronic search strategies to identify adverse effects data.J Med Libr Assoc. 2012 Apr;100(2):130-4. doi: 10.3163/1536-5050.100.2.012. J Med Libr Assoc. 2012. PMID: 22514510 Free PMC article. No abstract available.
References
-
- Tillman LA, Drake FM, Dixon JS, Wood JR. Review article: safety of bismuth in the treatment of gastrointestinal diseases. Aliment Pharmacol Ther. 1996;10:459–467. - PubMed
-
- Warren JR, Marshall BJ. Unidentified curved bacilli on gastric epithelium in active chronic gastritis. Lancet. 1983;321:1273–1275. - PubMed
-
- Wolle K, Malfertheiner P. Treatment of Helicobacter pylori. Best Pract Res Clin Gastroenterol. 2007;21:315–324. - PubMed
-
- Marshall BJ, Warren JR. Unidentified curved bacilli in the stomach of patients with gastritis and peptic ulceration. Lancet. 1984;1:1311–1315. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

