SOCS1 regulates the IFN but not NFkappaB pathway in TLR-stimulated human monocytes and macrophages
- PMID: 19017994
- PMCID: PMC3430718
- DOI: 10.4049/jimmunol.181.11.8018
SOCS1 regulates the IFN but not NFkappaB pathway in TLR-stimulated human monocytes and macrophages
Abstract
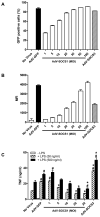
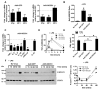
SOCS1 can regulate TLR-mediated signal transduction, yet mechanistic studies in murine macrophages have been confusing and contradictory. This study has used an adenoviral transfection system to determine the role of SOCS1 in the regulation of TNF-alpha production by activated human monocytes. Monocytes were infected with AdV-SOCS1 or with an empty vector control, AdV-GFP, for 24 h before activation with the TLR4 ligand, LPS. SOCS1 did not regulate TNF-alpha mRNA or protein production within the first two hours of TLR4 activation. However, SOCS1 suppressed the sustained production of TNF-alpha by primary human monocytes and synovial fluid macrophages ex vivo. In addition, SOCS1 regulated the production of IL-6, but not IL-10, by monocytes. Analysis of the early signaling pathway downstream of TLR4 demonstrated that SOCS1 had no regulatory effect on the activation or on the DNA binding capacity of NFkappaB. The late effects of LPS are mediated in part through the MyD88-independent pathway activating IRF3 and initiating the production of IFN-beta. In response to adenoviral infection and before LPS exposure, monocytes expressed enhanced levels of IFN-beta and Myxovirus A mRNA, an anti-viral molecule characterizing IFN-beta activity. These two genes were reduced in AdV-SOCS1-infected cells. Further, SOCS1 regulated IFN-dependent pathways in LPS-activated cells as evidenced by reduced IFN-beta production and STAT1 phosphorylation. Using AdV-infection to dissect SOCS1 control of IFN-dependent pathways, this study suggests that SOCS1-regulation of the IFN-dependent component of the LPS-induced TLR4 signaling pathway may contribute to the down-regulation of inflammatory cytokine production by AdV-SOCS1-infected human monocytes.
Figures
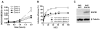

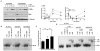

Similar articles
-
Endotoxin tolerance dysregulates MyD88- and Toll/IL-1R domain-containing adapter inducing IFN-beta-dependent pathways and increases expression of negative regulators of TLR signaling.J Leukoc Biol. 2009 Oct;86(4):863-75. doi: 10.1189/jlb.0309189. Epub 2009 Aug 5. J Leukoc Biol. 2009. PMID: 19656901 Free PMC article.
-
Inhibition of TLR4-induced IκB kinase activity by the RON receptor tyrosine kinase and its ligand, macrophage-stimulating protein.J Immunol. 2010 Dec 15;185(12):7309-16. doi: 10.4049/jimmunol.1000095. Epub 2010 Nov 15. J Immunol. 2010. PMID: 21078906 Free PMC article.
-
Peroxisome proliferator-activated receptor gamma negatively regulates IFN-beta production in Toll-like receptor (TLR) 3- and TLR4-stimulated macrophages by preventing interferon regulatory factor 3 binding to the IFN-beta promoter.J Biol Chem. 2011 Feb 18;286(7):5519-28. doi: 10.1074/jbc.M110.149823. Epub 2010 Dec 10. J Biol Chem. 2011. PMID: 21148557 Free PMC article.
-
IRF3 function and immunological gaps in sepsis.Front Immunol. 2024 Feb 5;15:1336813. doi: 10.3389/fimmu.2024.1336813. eCollection 2024. Front Immunol. 2024. PMID: 38375470 Free PMC article. Review.
-
Regulation of interferon and Toll-like receptor signaling during macrophage activation by opposing feedforward and feedback inhibition mechanisms.Immunol Rev. 2008 Dec;226:41-56. doi: 10.1111/j.1600-065X.2008.00707.x. Immunol Rev. 2008. PMID: 19161415 Free PMC article. Review.
Cited by
-
TLR8 Couples SOCS-1 and Restrains TLR7-Mediated Antiviral Immunity, Exacerbating West Nile Virus Infection in Mice.J Immunol. 2016 Dec 1;197(11):4425-4435. doi: 10.4049/jimmunol.1600902. Epub 2016 Oct 21. J Immunol. 2016. PMID: 27798161 Free PMC article.
-
Effects of Lansoprazole on the Lipopolysaccharide-Stimulated Toll-Like Receptor 4 Signal Transduction Systems: A Study Using the 293hTLR4/MD2-CD14 Cells.J Clin Biochem Nutr. 2009 Sep;45(2):241-7. doi: 10.3164/jcbn.09-42. Epub 2009 Aug 28. J Clin Biochem Nutr. 2009. PMID: 19794935 Free PMC article.
-
Intracellular delivery of a cell-penetrating SOCS1 that targets IFN-gamma signaling.Sci Signal. 2009 Jul 21;2(80):ra37. doi: 10.1126/scisignal.1162191. Sci Signal. 2009. PMID: 19622834 Free PMC article.
-
Role of position 627 of PB2 and the multibasic cleavage site of the hemagglutinin in the virulence of H5N1 avian influenza virus in chickens and ducks.PLoS One. 2012;7(2):e30960. doi: 10.1371/journal.pone.0030960. Epub 2012 Feb 21. PLoS One. 2012. PMID: 22363523 Free PMC article.
-
A respiratory syncytial virus persistent-infected cell line system reveals the involvement of SOCS1 in the innate antiviral response.Virol Sin. 2015 Jun;30(3):190-9. doi: 10.1007/s12250-015-3597-0. Epub 2015 Jun 19. Virol Sin. 2015. PMID: 26122642 Free PMC article.
References
-
- Huang Q, Ma Y, Adebayo A, Pope RM. Increased macrophage activation mediated through toll-like receptors in rheumatoid arthritis. Arthritis Rheum. 2007;56:2192–2201. - PubMed
-
- Akira S, Takeda K. Toll-like receptor signalling. Nat Rev Immunol. 2004;4:499–511. - PubMed
-
- O’Neill LA. How Toll-like receptors signal: what we know and what we don’t know. Curr Opin Immunol. 2006;18:3–9. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

