Surfactin-triggered small vesicle formation of negatively charged membranes: a novel membrane-lysis mechanism
- PMID: 18515378
- PMCID: PMC2553101
- DOI: 10.1529/biophysj.107.128322
Surfactin-triggered small vesicle formation of negatively charged membranes: a novel membrane-lysis mechanism
Abstract
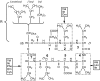
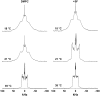
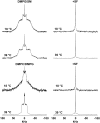
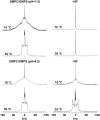
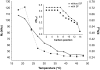
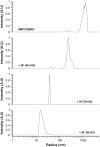
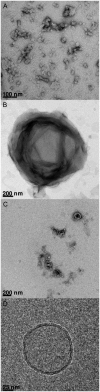
The molecular mode of action of the lipopeptide SF with zwitterionic and negatively charged model membranes has been investigated with solid-state NMR, light scattering, and electron microscopy. It has been found that this acidic lipopeptide (negatively charged) induces a strong destabilization of negatively charged micrometer-scale liposomes, leading to the formation of small unilamellar vesicles of a few 10s of nanometers. This transformation is detected for very low doses of SF (Ri = 200) and is complete for Ri = 50. The phenomenon has been observed for several membrane mixtures containing phosphatidylglycerol or phosphatidylserine. The vesicularization is not observed when the lipid negative charges are neutralized and a cholesterol-like effect is then evidenced, i.e., increase of gel membrane dynamics and decrease of fluid membrane microfluidity. The mechanism for small vesicle formation thus appears to be linked to severe changes in membrane curvature and could be described by a two-step action: 1), peptide insertion into membranes because of favorable van der Waals forces between the rather rigid cyclic and lipophilic part of SF and lipid chains and 2), electrostatic repulsion between like charges borne by lipid headgroups and the negatively charged SF amino acids. This might provide the basis for a novel mode of action of negatively charged lipopeptides.
Figures

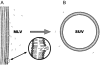
Similar articles
-
Molecular mechanism of membrane permeabilization by the peptide antibiotic surfactin.Biochim Biophys Acta. 2003 Apr 1;1611(1-2):91-7. doi: 10.1016/s0005-2736(03)00029-4. Biochim Biophys Acta. 2003. PMID: 12659949
-
Interaction of bee venom melittin with zwitterionic and negatively charged phospholipid bilayers: a spin-label electron spin resonance study.Biophys J. 1997 Feb;72(2 Pt 1):767-78. doi: 10.1016/s0006-3495(97)78711-3. Biophys J. 1997. PMID: 9017202 Free PMC article.
-
A study on the interactions of surfactin with phospholipid vesicles.Biochim Biophys Acta. 1999 May 12;1418(2):307-19. doi: 10.1016/s0005-2736(99)00039-5. Biochim Biophys Acta. 1999. PMID: 10320682
-
Effects of surfactin on membrane models displaying lipid phase separation.Biochim Biophys Acta. 2013 Feb;1828(2):801-15. doi: 10.1016/j.bbamem.2012.11.007. Epub 2012 Nov 14. Biochim Biophys Acta. 2013. PMID: 23159483
-
Role of Lipid Composition, Physicochemical Interactions, and Membrane Mechanics in the Molecular Actions of Microbial Cyclic Lipopeptides.J Membr Biol. 2019 Jun;252(2-3):131-157. doi: 10.1007/s00232-019-00067-4. Epub 2019 May 16. J Membr Biol. 2019. PMID: 31098678 Review.
Cited by
-
Vesicle Leakage Reflects the Target Selectivity of Antimicrobial Lipopeptides from Bacillus subtilis.Biophys J. 2015 Nov 17;109(10):2079-89. doi: 10.1016/j.bpj.2015.09.021. Biophys J. 2015. PMID: 26588567 Free PMC article.
-
Legionella pneumophila: The Paradox of a Highly Sensitive Opportunistic Waterborne Pathogen Able to Persist in the Environment.Front Microbiol. 2016 Apr 8;7:486. doi: 10.3389/fmicb.2016.00486. eCollection 2016. Front Microbiol. 2016. PMID: 27092135 Free PMC article. Review.
-
Digyalipopeptide A, an antiparasitic cyclic peptide from the Ghanaian Bacillus sp. strain DE2B.Beilstein J Org Chem. 2022 Dec 28;18:1763-1771. doi: 10.3762/bjoc.18.185. eCollection 2022. Beilstein J Org Chem. 2022. PMID: 36632531 Free PMC article.
-
Added Value of Biophysics to Study Lipid-Driven Biological Processes: The Case of Surfactins, a Class of Natural Amphiphile Molecules.Int J Mol Sci. 2022 Nov 10;23(22):13831. doi: 10.3390/ijms232213831. Int J Mol Sci. 2022. PMID: 36430318 Free PMC article. Review.
-
Lipopeptide production by Bacillus atrophaeus strain B44 and its biocontrol efficacy against cotton rhizoctoniosis.Biotechnol Lett. 2021 Jun;43(6):1183-1193. doi: 10.1007/s10529-021-03114-0. Epub 2021 Mar 18. Biotechnol Lett. 2021. PMID: 33738609
References
-
- Bulet, P., and R. Stöcklin. 2005. Insect antimicrobial peptides: structures, properties and gene regulation. Protein Pept. Lett. 12:3–11. - PubMed
-
- Pouny, Y., D. Rapaport, A. Mor, P. Nicolas, and Y. Shai. 1992. Interaction of antimicrobial dermaseptin and its fluorescently labeled analogues with phospholipid membranes. Biochemistry. 31:12416–12423. - PubMed
-
- Ehrenstein, G., and H. Lecar. 1977. Electrically gated ionic channels in lipid bilayers. Q. Rev. Biophys. 10:1–34. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

