Multiple forms of atypical rearrangements generating supernumerary derivative chromosome 15
- PMID: 18177502
- PMCID: PMC2249594
- DOI: 10.1186/1471-2156-9-2
Multiple forms of atypical rearrangements generating supernumerary derivative chromosome 15
Abstract
Background: Maternally-derived duplications that include the imprinted region on the proximal long arm of chromosome 15 underlie a complex neurobehavioral disorder characterized by cognitive impairment, seizures and a substantial risk for autism spectrum disorders1. The duplications most often take the form of a supernumerary pseudodicentric derivative chromosome 15 [der(15)] that has been called inverted duplication 15 or isodicentric 15 [idic(15)], although interstitial rearrangements also occur. Similar to the deletions found in most cases of Angelman and Prader Willi syndrome, the duplications appear to be mediated by unequal homologous recombination involving low copy repeats (LCR) that are found clustered in the region. Five recurrent breakpoints have been described in most cases of segmental aneuploidy of chromosome 15q11-q13 and previous studies have shown that most idic(15) chromosomes arise through BP3:BP3 or BP4:BP5 recombination events.
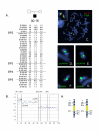
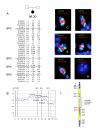
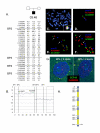
Results: Here we describe four duplication chromosomes that show evidence of atypical recombination events that involve regions outside the common breakpoints. Additionally, in one patient with a mosaic complex der(15), we examined homologous pairing of chromosome 15q11-q13 alleles by FISH in a region of frontal cortex, which identified mosaicism in this tissue and also demonstrated pairing of the signals from the der(15) and the normal homologues.
Conclusion: Involvement of atypical BP in the generation of idic(15) chromosomes can lead to considerable structural heterogeneity.
Figures
Similar articles
-
Large genomic duplicons map to sites of instability in the Prader-Willi/Angelman syndrome chromosome region (15q11-q13).Hum Mol Genet. 1999 Jun;8(6):1025-37. doi: 10.1093/hmg/8.6.1025. Hum Mol Genet. 1999. PMID: 10332034
-
Screening for genomic rearrangements and methylation abnormalities of the 15q11-q13 region in autism spectrum disorders.Biol Psychiatry. 2009 Aug 15;66(4):349-59. doi: 10.1016/j.biopsych.2009.01.025. Epub 2009 Mar 17. Biol Psychiatry. 2009. PMID: 19278672
-
Prader-Willi, Angelman, and 15q11-q13 Duplication Syndromes.Pediatr Clin North Am. 2015 Jun;62(3):587-606. doi: 10.1016/j.pcl.2015.03.004. Epub 2015 Apr 22. Pediatr Clin North Am. 2015. PMID: 26022164 Free PMC article. Review.
-
Parental and chromosomal origins of microdeletion and duplication syndromes involving 7q11.23, 15q11-q13 and 22q11.Eur J Hum Genet. 2006 Jul;14(7):831-7. doi: 10.1038/sj.ejhg.5201617. Epub 2006 Apr 12. Eur J Hum Genet. 2006. PMID: 16617304
-
Duplication within chromosome region 15q11-q13 in a patient with similarities to Prader-Willi syndrome confirmed by region-specific and band-specific fish.Genet Couns. 1999;10(2):123-32. Genet Couns. 1999. PMID: 10422004 Review.
Cited by
-
Autistic disorder associated with a paternally derived unbalanced translocation leading to duplication of chromosome 15pter-q13.2: a case report.Mol Cytogenet. 2009 Dec 18;2:27. doi: 10.1186/1755-8166-2-27. Mol Cytogenet. 2009. PMID: 20021661 Free PMC article.
-
Possible Phenotypic Consequences of Structural Differences in Idic(15) in a Small Cohort of Patients.Int J Mol Sci. 2019 Oct 5;20(19):4935. doi: 10.3390/ijms20194935. Int J Mol Sci. 2019. PMID: 31590400 Free PMC article.
-
The inv dup (15) or idic (15) syndrome (Tetrasomy 15q).Orphanet J Rare Dis. 2008 Nov 19;3:30. doi: 10.1186/1750-1172-3-30. Orphanet J Rare Dis. 2008. PMID: 19019226 Free PMC article. Review.
-
Hominoid chromosomal rearrangements on 17q map to complex regions of segmental duplication.Genome Biol. 2008;9(2):R28. doi: 10.1186/gb-2008-9-2-r28. Epub 2008 Feb 7. Genome Biol. 2008. PMID: 18257913 Free PMC article.
-
[A clinical and genetic analysis of a child with supernumerary marker chromosome 15-caused mental retardation, intractable epilepsy, and central precocious puberty].Zhongguo Dang Dai Er Ke Za Zhi. 2018 Aug;20(8):652-657. doi: 10.7499/j.issn.1008-8830.2018.08.010. Zhongguo Dang Dai Er Ke Za Zhi. 2018. PMID: 30111475 Free PMC article. Chinese.
References
-
- Battaglia A, Gurrieri F, Bertini E, Bellacosa A, Pomponi MG, Paravatou-Petsotas M, Mazza S, Neri G. The inv dup(15) syndrome: a clinically recognizable syndrome with altered behavior, mental retardation, and epilepsy. Neurology. 1997;48:1081–1086. - PubMed
-
- Amos-Landgraf JM, Ji Y, Gottlieb W, Depinet T, Wandstrat AE, Cassidy SB, Driscoll DJ, Rogan PK, Schwartz S, Nicholls RD. Chromosome breakage in the Prader-Willi and Angelman syndromes involves recombination between large, transcribed repeats at proximal and distal breakpoints. Am J Hum Genet. 1999;65:370–386. doi: 10.1086/302510. - DOI - PMC - PubMed
-
- Sahoo T, Bacino CA, German JR, Shaw CA, Bird LM, Kimoninis V, Anselm I, Beaudet AL, Peters SU. Identification of novel deletions of 15q11q13 in Angelman syndrome by array-CGH: molecular characterization and genotype-phenotype correlations. European journal of human genetics. 2007;15:943–949. doi: 10.1038/sj.ejhg.5201859. - DOI - PubMed

