TASK-3 two-pore domain potassium channels enable sustained high-frequency firing in cerebellar granule neurons
- PMID: 17728447
- PMCID: PMC6673138
- DOI: 10.1523/JNEUROSCI.1427-07.2007
TASK-3 two-pore domain potassium channels enable sustained high-frequency firing in cerebellar granule neurons
Abstract
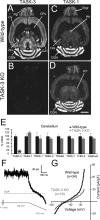
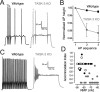
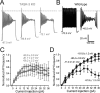
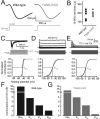
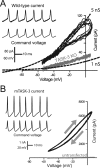
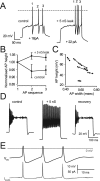
The ability of neurons, such as cerebellar granule neurons (CGNs), to fire action potentials (APs) at high frequencies during sustained depolarization is usually explained in relation to the functional properties of voltage-gated ion channels. Two-pore domain potassium (K(2P)) channels are considered to simply hyperpolarize the resting membrane potential (RMP) by increasing the potassium permeability of the membrane. However, we find that CGNs lacking the TASK-3 type K(2P) channel exhibit marked accommodation of action potential firing. The accommodation phenotype was not associated with any change in the functional properties of the underlying voltage-gated sodium channels, nor could it be explained by the more depolarized RMP that resulted from TASK-3 channel deletion. A functional rescue, involving the introduction of a nonlinear leak conductance with a dynamic current clamp, was able to restore wild-type firing properties to adult TASK-3 knock-out CGNs. Thus, in addition to the accepted role of TASK-3 channels in limiting neuronal excitability, by increasing the resting potassium conductance TASK-3 channels also increase excitability by supporting high-frequency firing once AP threshold is reached.
Figures


Similar articles
-
Identification of native rat cerebellar granule cell currents due to background K channel KCNK5 (TASK-2).Brain Res Mol Brain Res. 2004 Sep 28;128(2):112-20. doi: 10.1016/j.molbrainres.2004.06.007. Brain Res Mol Brain Res. 2004. PMID: 15363886
-
TASK channels contribute to the K+-dominated leak current regulating respiratory rhythm generation in vitro.J Neurosci. 2010 Mar 24;30(12):4273-84. doi: 10.1523/JNEUROSCI.4017-09.2010. J Neurosci. 2010. PMID: 20335463 Free PMC article.
-
Voltage-gated potassium channels activated during action potentials in layer V neocortical pyramidal neurons.J Neurophysiol. 2000 Jan;83(1):70-80. doi: 10.1152/jn.2000.83.1.70. J Neurophysiol. 2000. PMID: 10634854
-
Two-pore domain potassium channels enable action potential generation in the absence of voltage-gated potassium channels.Pflugers Arch. 2015 May;467(5):989-99. doi: 10.1007/s00424-014-1660-6. Epub 2014 Dec 9. Pflugers Arch. 2015. PMID: 25482670 Free PMC article. Review.
-
Voltage-gated sodium currents in cerebellar Purkinje neurons: functional and molecular diversity.Cell Mol Life Sci. 2018 Oct;75(19):3495-3505. doi: 10.1007/s00018-018-2868-y. Epub 2018 Jul 7. Cell Mol Life Sci. 2018. PMID: 29982847 Free PMC article. Review.
Cited by
-
17β-estradiol downregulated the expression of TASK-1 channels in mouse neuroblastoma N2A cells.J Membr Biol. 2014 Mar;247(3):273-9. doi: 10.1007/s00232-014-9632-5. Epub 2014 Jan 17. J Membr Biol. 2014. PMID: 24435466
-
Increased excitability and altered action potential waveform in cerebellar granule neurons of the Ts65Dn mouse model of Down syndrome.Brain Res. 2012 Jul 17;1465(8):10-7. doi: 10.1016/j.brainres.2012.05.027. Epub 2012 May 22. Brain Res. 2012. PMID: 22627164 Free PMC article.
-
Biophysical properties of human medulloblastoma cells.J Membr Biol. 2010 Oct;237(2-3):59-69. doi: 10.1007/s00232-010-9306-x. Epub 2010 Oct 8. J Membr Biol. 2010. PMID: 20931182
-
TASK channels determine pH sensitivity in select respiratory neurons but do not contribute to central respiratory chemosensitivity.J Neurosci. 2007 Dec 19;27(51):14049-58. doi: 10.1523/JNEUROSCI.4254-07.2007. J Neurosci. 2007. PMID: 18094244 Free PMC article.
-
An unexpected role for TASK-3 potassium channels in network oscillations with implications for sleep mechanisms and anesthetic action.Proc Natl Acad Sci U S A. 2009 Oct 13;106(41):17546-51. doi: 10.1073/pnas.0907228106. Epub 2009 Sep 24. Proc Natl Acad Sci U S A. 2009. PMID: 19805135 Free PMC article.
References
-
- Aller MI, Veale E, Linden AM, Sandu C, Schwaninger M, Evans L, Korpi ER, Mathie A, Wisden W, Brickley SG. Modifying the subunit composition of TASK channels alters the modulation of a leak conductance in cerebellar granule neurons and is associated with impaired motor performance. J Neurosci. 2005;25:11455–11467. - PMC - PubMed
-
- Bean BP. The action potential in mammalian central neurons. Nat Rev Neurosci. 2007;8:451–465. - PubMed
-
- Berg AP, Bayliss DA. Striatal cholinergic interneurons express a receptor-insensitive homomeric TASK-3-like background K+ current. J Neurophysiol. 2007;97:1546–1552. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous
