The EF-G-like GTPase Snu114p regulates spliceosome dynamics mediated by Brr2p, a DExD/H box ATPase
- PMID: 16885028
- PMCID: PMC3777414
- DOI: 10.1016/j.molcel.2006.05.043
The EF-G-like GTPase Snu114p regulates spliceosome dynamics mediated by Brr2p, a DExD/H box ATPase
Abstract
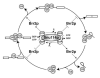
Binding of a pre-mRNA substrate triggers spliceosome activation, whereas the release of the mRNA product triggers spliceosome disassembly. The mechanisms that underlie the regulation of these rearrangements remain unclear. We find evidence that the GTPase Snu114p mediates the regulation of spliceosome activation and disassembly. Specifically, both unwinding of U4/U6, required for spliceosome activation, and disassembly of the postsplicing U2/U6.U5.intron complex are repressed by Snu114p bound to GDP and derepressed by Snu114p bound to GTP or nonhydrolyzable GTP analogs. Further, similar to U4/U6 unwinding, spliceosome disassembly requires the DExD/H box ATPase Brr2p. Together, our data define a common mechanism for regulating and executing spliceosome activation and disassembly. Although sequence similarity with EF-G suggests Snu114p functions as a molecular motor, our findings indicate that Snu114p functions as a classic regulatory G protein. We propose that Snu114p serves as a signal-dependent switch that transduces signals to Brr2p to control spliceosome dynamics.
Figures
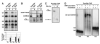

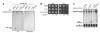

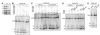
Similar articles
-
Analysis of synthetic lethality reveals genetic interactions between the GTPase Snu114p and snRNAs in the catalytic core of the Saccharomyces cerevisiae spliceosome.Genetics. 2009 Oct;183(2):497-515-1SI-4SI. doi: 10.1534/genetics.109.107243. Epub 2009 Jul 20. Genetics. 2009. PMID: 19620389 Free PMC article.
-
Mutagenesis suggests several roles of Snu114p in pre-mRNA splicing.J Biol Chem. 2003 Jul 25;278(30):28324-34. doi: 10.1074/jbc.M303043200. Epub 2003 May 7. J Biol Chem. 2003. PMID: 12736260
-
Functional contacts with a range of splicing proteins suggest a central role for Brr2p in the dynamic control of the order of events in spliceosomes of Saccharomyces cerevisiae.Genetics. 2001 Apr;157(4):1451-67. doi: 10.1093/genetics/157.4.1451. Genetics. 2001. PMID: 11290703 Free PMC article.
-
RNA Splicing by the Spliceosome.Annu Rev Biochem. 2020 Jun 20;89:359-388. doi: 10.1146/annurev-biochem-091719-064225. Epub 2019 Dec 3. Annu Rev Biochem. 2020. PMID: 31794245 Review.
-
How Is Precursor Messenger RNA Spliced by the Spliceosome?Annu Rev Biochem. 2020 Jun 20;89:333-358. doi: 10.1146/annurev-biochem-013118-111024. Epub 2019 Dec 9. Annu Rev Biochem. 2020. PMID: 31815536 Review.
Cited by
-
Visualizing the splicing of single pre-mRNA molecules in whole cell extract.RNA. 2008 Jan;14(1):170-9. doi: 10.1261/rna.794808. Epub 2007 Nov 19. RNA. 2008. PMID: 18025254 Free PMC article.
-
Spliceosome discards intermediates via the DEAH box ATPase Prp43p.Proc Natl Acad Sci U S A. 2010 Jun 1;107(22):10020-5. doi: 10.1073/pnas.0906022107. Epub 2010 May 12. Proc Natl Acad Sci U S A. 2010. PMID: 20463285 Free PMC article.
-
Insights into the activation of the helicase Prp43 by biochemical studies and structural mass spectrometry.Nucleic Acids Res. 2014 Jan;42(2):1162-79. doi: 10.1093/nar/gkt985. Epub 2013 Oct 27. Nucleic Acids Res. 2014. PMID: 24165877 Free PMC article.
-
A snRNP's ordered path to maturity.Genes Dev. 2011 Aug 1;25(15):1563-7. doi: 10.1101/gad.17311211. Genes Dev. 2011. PMID: 21828266 Free PMC article.
-
Spliceosome structure and function.Cold Spring Harb Perspect Biol. 2011 Jul 1;3(7):a003707. doi: 10.1101/cshperspect.a003707. Cold Spring Harb Perspect Biol. 2011. PMID: 21441581 Free PMC article. Review.
References
-
- Achsel T, Ahrens K, Brahms H, Teigelkamp S, Lührmann R. The human U5-220kD protein (hPrp8) forms a stable RNA-free complex with several U5-specific proteins, including an RNA unwindase, a homologue of ribosomal elongation factor EF-2, and a novel WD-40 protein. Mol. Cell. Biol. 1998;18:6756–6766. - PMC - PubMed
-
- Bartels C, Urlaub H, Lührmann R, Fabrizio P. Mutagenesis suggests several roles of Snu114p in pre-mRNA splicing. J. Biol. Chem. 2003;278:28324–28334. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

