MR characteristics and neuropathology in adult-onset autosomal dominant leukodystrophy with autonomic symptoms
- PMID: 16611789
- PMCID: PMC8133955
MR characteristics and neuropathology in adult-onset autosomal dominant leukodystrophy with autonomic symptoms
Abstract
Background and purpose: Three families with adult-onset autosomal dominant leukodystrophy (ADLD) presenting autonomic dysfunction as the first symptom are reported. We describe detailed MR appearances of the brain in 2 new families and neuropathology in 2 patients and compare the findings with those in other adult-onset leukodystrophies.
Methods: Twenty subjects (12 women and 8 men; age range, 29-70 years) from 2 unrelated families with ADLD were examined with MR. Six subjects were asymptomatic. Fourteen had autonomic dysfunction. Eleven of them also had pyramidal signs and ataxia. The brains of 2 autopsied patients were examined histopathologically.
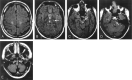
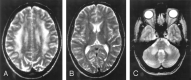
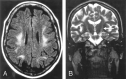
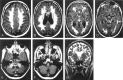
Results: Two subjects manifested no neurologic symptoms, signs, or MR pathology. Eighteen subjects displayed radiologic abnormalities ranging from subtle T2 high-signal-intensity changes in the upper corticospinal tract to extensive confluent white matter changes, predominantly in a frontoparietal distribution, along the corticospinal tracts down to the medulla oblongata and in the upper and middle cerebellar peduncles. Periventricular white matter was spared or less affected than the adjacent white matter. Histopathology revealed marked loss of cerebral and cerebellar myelin without signs of inflammation. Oligodendrocytes were relatively spared, the number of axons not markedly decreased, and reactive gliosis was modest. The number of Purkinje cells in the cerebellum was reduced.
Conclusions: Two families with adult-onset ADLD with the disease entity originally reported by Eldridge et al. (N Engl J Med 1984;311:948-53) were described. We propose naming the disease "adult-onset ADLD with autonomic symptoms." The characteristic radiologic findings, combined with the clinical symptoms and mode of inheritance, enable the diagnosis.
Figures

Similar articles
-
Genomic duplications mediate overexpression of lamin B1 in adult-onset autosomal dominant leukodystrophy (ADLD) with autonomic symptoms.Neurogenetics. 2011 Feb;12(1):65-72. doi: 10.1007/s10048-010-0269-y. Epub 2011 Jan 12. Neurogenetics. 2011. PMID: 21225301
-
MR imaging characteristics and neuropathology of the spinal cord in adult-onset autosomal dominant leukodystrophy with autonomic symptoms.AJNR Am J Neuroradiol. 2009 Feb;30(2):328-35. doi: 10.3174/ajnr.A1354. Epub 2008 Oct 22. AJNR Am J Neuroradiol. 2009. PMID: 18945794 Free PMC article.
-
¹H-MR spectroscopy of adult-onset autosomal dominant leukodystrophy with autonomic symptoms.Neuroradiology. 2013 Aug;55(8):933-939. doi: 10.1007/s00234-013-1174-5. Epub 2013 May 1. Neuroradiology. 2013. PMID: 23636437
-
[A Japanese family with probably autosomal dominant adult-onset leukodystrophy].Rinsho Shinkeigaku. 1996 Aug;36(8):968-72. Rinsho Shinkeigaku. 1996. PMID: 8958750 Review. Japanese.
-
Globoid cell leukodystrophy: distinguishing early-onset from late-onset disease using a brain MR imaging scoring method.AJNR Am J Neuroradiol. 1999 Feb;20(2):316-23. AJNR Am J Neuroradiol. 1999. PMID: 10094363 Free PMC article. Review.
Cited by
-
Nuclear lamins in the brain - new insights into function and regulation.Mol Neurobiol. 2013 Feb;47(1):290-301. doi: 10.1007/s12035-012-8350-1. Epub 2012 Oct 14. Mol Neurobiol. 2013. PMID: 23065386 Free PMC article. Review.
-
LMNB1-Related Adult-Onset Autosomal Dominant Leukodystrophy Presenting as Movement Disorder: A Case Report and Review of the Literature.Front Neurosci. 2019 Oct 21;13:1030. doi: 10.3389/fnins.2019.01030. eCollection 2019. Front Neurosci. 2019. PMID: 31695592 Free PMC article.
-
Genomic duplications mediate overexpression of lamin B1 in adult-onset autosomal dominant leukodystrophy (ADLD) with autonomic symptoms.Neurogenetics. 2011 Feb;12(1):65-72. doi: 10.1007/s10048-010-0269-y. Epub 2011 Jan 12. Neurogenetics. 2011. PMID: 21225301
-
Adult-onset autosomal dominant leukodystrophy: linking nuclear envelope to myelin.J Neurosci. 2011 Jan 26;31(4):1163-6. doi: 10.1523/JNEUROSCI.5994-10.2011. J Neurosci. 2011. PMID: 21273400 Free PMC article. Review. No abstract available.
-
Analysis of LMNB1 duplications in autosomal dominant leukodystrophy provides insights into duplication mechanisms and allele-specific expression.Hum Mutat. 2013 Aug;34(8):1160-71. doi: 10.1002/humu.22348. Epub 2013 May 28. Hum Mutat. 2013. PMID: 23649844 Free PMC article.
References
-
- Berger J, Moser HW, Forss-Petter S. Leukodystrophies: recent developments in genetics, molecular biology, pathogenesis and treatment. Curr Opin Neurol 2001;14:305–12 - PubMed
-
- McKusick-Nathans Institute for Genetic Medicine, Johns Hopkins University and National Center for Biotechnology Information, National Library of Medicine. Online Mendelian inheritance in man, OMIM.2000. . Available at http://www.ncbi.nlm.nih.gov/entrez/query.fcgi?db=OMIM/.
-
- Eldridge R, Anayiotos CP, Schlesinger S, et al. Hereditary adult-onset leukodystrophy simulating chronic progressive multiple sclerosis. N Engl J Med 1984;311:948–53 - PubMed
-
- Schwankhaus JD, Patronas N, Dorwart R, et al. Computed tomography and magnetic resonance imaging in adult-onset leukodystrophy. Arch Neurol 1988;45:1004–08 - PubMed
-
- Schwankhaus JD, Katz DA, Eldridge R, et al. Clinical and pathological features of an autosomal dominant, adult-onset leukodystrophy simulating chronic progressive multiple sclerosis. Arch Neurol 1994;51:757–66 - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials
