Mutation of the LUNATIC FRINGE gene in humans causes spondylocostal dysostosis with a severe vertebral phenotype
- PMID: 16385447
- PMCID: PMC1380221
- DOI: 10.1086/498879
Mutation of the LUNATIC FRINGE gene in humans causes spondylocostal dysostosis with a severe vertebral phenotype
Abstract
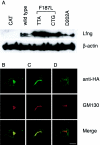
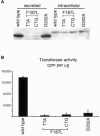
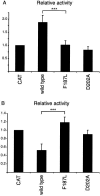
The spondylocostal dysostoses (SCDs) are a heterogeneous group of vertebral malsegmentation disorders that arise during embryonic development by a disruption of somitogenesis. Previously, we had identified two genes that cause a subset of autosomal recessive forms of this disease: DLL3 (SCD1) and MESP2 (SCD2). These genes are important components of the Notch signaling pathway, which has multiple roles in development and disease. Here, we have used a candidate-gene approach to identify a mutation in a third Notch pathway gene, LUNATIC FRINGE (LFNG), in a family with autosomal recessive SCD. LFNG encodes a glycosyltransferase that modifies the Notch family of cell-surface receptors, a key step in the regulation of this signaling pathway. A missense mutation was identified in a highly conserved phenylalanine close to the active site of the enzyme. Functional analysis revealed that the mutant LFNG was not localized to the correct compartment of the cell, was unable to modulate Notch signaling in a cell-based assay, and was enzymatically inactive. This represents the first known mutation in the human LFNG gene and reinforces the hypothesis that proper regulation of the Notch signaling pathway is an absolute requirement for the correct patterning of the axial skeleton.
Figures


Similar articles
-
Mutation of Hairy-and-Enhancer-of-Split-7 in humans causes spondylocostal dysostosis.Hum Mol Genet. 2008 Dec 1;17(23):3761-6. doi: 10.1093/hmg/ddn272. Epub 2008 Sep 5. Hum Mol Genet. 2008. PMID: 18775957
-
Autosomal dominant spondylocostal dysostosis in three generations of a Macedonian family: Negative mutation analysis of DLL3, MESP2, HES7, and LFNG.Am J Med Genet A. 2010 Jun;152A(6):1378-82. doi: 10.1002/ajmg.a.33471. Am J Med Genet A. 2010. PMID: 20503311
-
Mutation of the fucose-specific beta1,3 N-acetylglucosaminyltransferase LFNG results in abnormal formation of the spine.Biochim Biophys Acta. 2009 Feb;1792(2):100-11. doi: 10.1016/j.bbadis.2008.11.003. Epub 2008 Nov 13. Biochim Biophys Acta. 2009. PMID: 19061953 Review.
-
Mutations in the human delta homologue, DLL3, cause axial skeletal defects in spondylocostal dysostosis.Nat Genet. 2000 Apr;24(4):438-41. doi: 10.1038/74307. Nat Genet. 2000. PMID: 10742114
-
Defective somitogenesis and abnormal vertebral segmentation in man.Adv Exp Med Biol. 2008;638:164-89. doi: 10.1007/978-0-387-09606-3_9. Adv Exp Med Biol. 2008. PMID: 21038776 Review.
Cited by
-
Molecular genetics of congenital diaphragmatic defects.Ann Med. 2007;39(4):261-74. doi: 10.1080/07853890701326883. Ann Med. 2007. PMID: 17558598 Free PMC article. Review.
-
Notch signaling pathway: architecture, disease, and therapeutics.Signal Transduct Target Ther. 2022 Mar 24;7(1):95. doi: 10.1038/s41392-022-00934-y. Signal Transduct Target Ther. 2022. PMID: 35332121 Free PMC article. Review.
-
In vitro characterization of the human segmentation clock.Nature. 2020 Apr;580(7801):113-118. doi: 10.1038/s41586-019-1885-9. Epub 2020 Jan 8. Nature. 2020. PMID: 31915384 Free PMC article.
-
Notch and the skeleton.Mol Cell Biol. 2010 Feb;30(4):886-96. doi: 10.1128/MCB.01285-09. Epub 2009 Dec 7. Mol Cell Biol. 2010. PMID: 19995916 Free PMC article. Review.
-
Fucose as a nutrient ligand for Dikarya and a building block of early diverging lineages.IMA Fungus. 2023 Sep 5;14(1):17. doi: 10.1186/s43008-023-00123-8. IMA Fungus. 2023. PMID: 37670396 Free PMC article.
References
Web Resources
-
- Online Mendelian Inheritance in Man (OMIM), http://www.ncbi.nlm.nih.gov/Omim/ (for SCD1 and SCD2)
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Molecular Biology Databases

