Crystallographic comparison of manganese- and iron-dependent homoprotocatechuate 2,3-dioxygenases
- PMID: 15028678
- PMCID: PMC374394
- DOI: 10.1128/JB.186.7.1945-1958.2004
Crystallographic comparison of manganese- and iron-dependent homoprotocatechuate 2,3-dioxygenases
Abstract
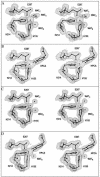
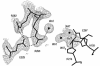
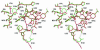
The X-ray crystal structures of homoprotocatechuate 2,3-dioxygenases isolated from Arthrobacter globiformis and Brevibacterium fuscum have been determined to high resolution. These enzymes exhibit 83% sequence identity, yet their activities depend on different transition metals, Mn2+ and Fe2+, respectively. The structures allow the origins of metal ion selectivity and aspects of the molecular mechanism to be examined in detail. The homotetrameric enzymes belong to the type I family of extradiol dioxygenases (vicinal oxygen chelate superfamily); each monomer has four betaalphabetabetabeta modules forming two structurally homologous N-terminal and C-terminal barrel-shaped domains. The active-site metal is located in the C-terminal barrel and is ligated by two equatorial ligands, H214NE1 and E267OE1; one axial ligand, H155NE1; and two to three water molecules. The first and second coordination spheres of these enzymes are virtually identical (root mean square difference over all atoms, 0.19 A), suggesting that the metal selectivity must be due to changes at a significant distance from the metal and/or changes that occur during folding. The substrate (2,3-dihydroxyphenylacetate [HPCA]) chelates the metal asymmetrically at sites trans to the two imidazole ligands and interacts with a unique, mobile C-terminal loop. The loop closes over the bound substrate, presumably to seal the active site as the oxygen activation process commences. An "open" coordination site trans to E267 is the likely binding site for O2. The geometry of the enzyme-substrate complexes suggests that if a transiently formed metal-superoxide complex attacks the substrate without dissociation from the metal, it must do so at the C-3 position. Second-sphere active-site residues that are positioned to interact with the HPCA and/or bound O2 during catalysis are identified and discussed in the context of current mechanistic hypotheses.
Figures

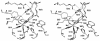

Similar articles
-
Cloning, overexpression, and mutagenesis of the gene for homoprotocatechuate 2,3-dioxygenase from Brevibacterium fuscum.Protein Expr Purif. 1997 Jun;10(1):1-9. doi: 10.1006/prep.1996.0703. Protein Expr Purif. 1997. PMID: 9179284
-
Manganese(II) active site mutants of 3,4-dihydroxyphenylacetate 2,3-dioxygenase from Arthrobacter globiformis strain CM-2.Biochemistry. 1997 Feb 25;36(8):2147-53. doi: 10.1021/bi962362i. Biochemistry. 1997. PMID: 9047314
-
4-nitrocatechol as a probe of a Mn(II)-dependent extradiol-cleaving catechol dioxygenase (MndD): comparison with relevant Fe(II) and Mn(II) model complexes.J Biol Inorg Chem. 2003 Feb;8(3):263-72. doi: 10.1007/s00775-002-0411-x. Epub 2002 Nov 9. J Biol Inorg Chem. 2003. PMID: 12589562
-
A two-electron-shell game: intermediates of the extradiol-cleaving catechol dioxygenases.J Biol Inorg Chem. 2014 Jun;19(4-5):491-504. doi: 10.1007/s00775-014-1122-9. Epub 2014 Mar 11. J Biol Inorg Chem. 2014. PMID: 24615282 Free PMC article. Review.
-
Structural and mechanistic comparisons of the metal-binding members of the vicinal oxygen chelate (VOC) superfamily.J Inorg Biochem. 2011 Oct;105(10):1259-72. doi: 10.1016/j.jinorgbio.2011.06.006. Epub 2011 Jul 4. J Inorg Biochem. 2011. PMID: 21820381 Review.
Cited by
-
Substrate-mediated oxygen activation by homoprotocatechuate 2,3-dioxygenase: intermediates formed by a tyrosine 257 variant.Biochemistry. 2012 Nov 6;51(44):8743-54. doi: 10.1021/bi301114x. Epub 2012 Oct 29. Biochemistry. 2012. PMID: 23066705 Free PMC article.
-
Metallation and mismetallation of iron and manganese proteins in vitro and in vivo: the class I ribonucleotide reductases as a case study.Metallomics. 2012 Oct;4(10):1020-36. doi: 10.1039/c2mt20142a. Epub 2012 Sep 18. Metallomics. 2012. PMID: 22991063 Free PMC article. Review.
-
Enzyme Substrate Complex of the H200C Variant of Homoprotocatechuate 2,3-Dioxygenase: Mössbauer and Computational Studies.Inorg Chem. 2016 Jun 20;55(12):5862-70. doi: 10.1021/acs.inorgchem.6b00148. Epub 2016 Jun 8. Inorg Chem. 2016. PMID: 27275865 Free PMC article.
-
Nuclear Resonance Vibrational Spectroscopy Definition of Peroxy Intermediates in Catechol Dioxygenases: Factors that Determine Extra- versus Intradiol Cleavage.J Am Chem Soc. 2023 Jul 19;145(28):15230-15250. doi: 10.1021/jacs.3c02242. Epub 2023 Jul 6. J Am Chem Soc. 2023. PMID: 37414058 Free PMC article.
-
Characterization of an O2 adduct of an active cobalt-substituted extradiol-cleaving catechol dioxygenase.J Am Chem Soc. 2012 Jan 18;134(2):796-9. doi: 10.1021/ja2095365. Epub 2011 Dec 29. J Am Chem Soc. 2012. PMID: 22175783 Free PMC article.
References
-
- Arciero, D. M., and J. D. Lipscomb. 1986. Binding of 17O-labeled substrate and inhibitors to protocatechuate 4,5-dioxygenase-nitrosyl complex. Evidence for direct substrate binding to the active site Fe2+ of extradiol dioxygenases. J. Biol. Chem. 261:2170-2178. - PubMed
-
- Arciero, D. M., J. D. Lipscomb, B. H. Huynh, T. A. Kent, and E. Münck. 1983. EPR and Mössbauer studies of protocatechuate 4,5-dioxygenase. Characterization of a new Fe2+ environment. J. Biol. Chem. 258:14981-14991. - PubMed
-
- Arciero, D. M., A. M. Orville, and J. D. Lipscomb. 1985. [17O]water and nitric oxide binding by protocatechuate 4,5-dioxygenase and catechol 2,3-dioxygenase. Evidence for binding of exogenous ligands to the active site Fe2+ of extradiol dioxygenases. J. Biol. Chem. 260:14035-14044. - PubMed
-
- Armstrong, R. N. 2000. Mechanistic diversity in a metalloenzyme superfamily. Biochemistry 39:13625-13632. - PubMed
-
- Asturias, J. A., L. D. Eltis, M. Prucha, and K. N. Timmis. 1994. Analysis of three 2,3-dihydroxybiphenyl 1,2-dioxygenases found in Rhodococcus globerulus P6. Identification of a new family of extradiol dioxygenases. J. Biol. Chem. 269:7807-7815. - PubMed
Publication types
MeSH terms
Substances
Associated data
- Actions
- Actions
- Actions
- Actions
- Actions
- Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

