Acid-responsive gene induction of ammonia-producing enzymes in Helicobacter pylori is mediated via a metal-responsive repressor cascade
- PMID: 14742519
- PMCID: PMC321643
- DOI: 10.1128/IAI.72.2.766-773.2004
Acid-responsive gene induction of ammonia-producing enzymes in Helicobacter pylori is mediated via a metal-responsive repressor cascade
Abstract
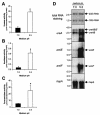
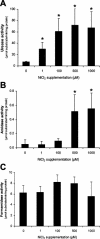
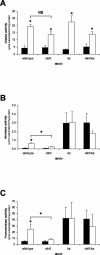
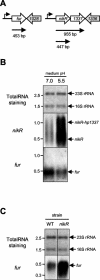
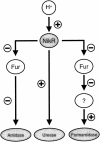
Although the adaptive mechanisms allowing the gastric pathogen Helicobacter pylori to survive acid shocks have been well documented, the mechanisms allowing growth at mildly acidic conditions (pH approximately 5.5) are still poorly understood. Here we demonstrate that H. pylori strain 26695 increases the transcription and activity of its urease, amidase, and formamidase enzymes four- to ninefold in response to growth at pH 5.5. Supplementation of growth medium with NiCl2 resulted in a similar induction of urease activity (at low NiCl2 concentration) and amidase activity (at > or = 500 micro M NiCl2) but did not affect formamidase activity. Mutation of the fur gene, which encodes an iron-responsive repressor of both amidases, resulted in a constitutively high level of amidase and formamidase activity at either pH but did not affect urease activity at pH 7.0 or pH 5.5. In contrast, mutation of the nikR gene, encoding the nickel-responsive activator of urease expression, resulted in a significant reduction of acid-responsive induction of amidase and formamidase activity. Finally, acid-responsive repression of fur transcription was absent in the H. pylori nikR mutant, whereas transcription of the nikR gene itself was increased at pH 5.5 in wild-type H. pylori. We hypothesize that H. pylori uses a repressor cascade to respond to low pH, with NikR initiating the response directly via the urease operon and indirectly via the members of the Fur regulon.
Figures
Similar articles
-
The nickel-responsive regulator NikR controls activation and repression of gene transcription in Helicobacter pylori.Infect Immun. 2005 Nov;73(11):7252-8. doi: 10.1128/IAI.73.11.7252-7258.2005. Infect Immun. 2005. PMID: 16239520 Free PMC article.
-
Differential regulation of amidase- and formamidase-mediated ammonia production by the Helicobacter pylori fur repressor.J Biol Chem. 2003 Mar 14;278(11):9052-7. doi: 10.1074/jbc.M207542200. Epub 2002 Dec 23. J Biol Chem. 2003. PMID: 12499381
-
NikR mediates nickel-responsive transcriptional induction of urease expression in Helicobacter pylori.Infect Immun. 2002 Jun;70(6):2846-52. doi: 10.1128/IAI.70.6.2846-2852.2002. Infect Immun. 2002. PMID: 12010971 Free PMC article.
-
Acid-responsive gene regulation in the human pathogen Helicobacter pylori.J Biotechnol. 2006 Oct 20;126(1):52-60. doi: 10.1016/j.jbiotec.2006.03.045. Epub 2006 May 19. J Biotechnol. 2006. PMID: 16713649 Review.
-
Multiple regulatory mechanisms for pH homeostasis in the gastric pathogen, Helicobacter pylori.Adv Genet. 2022;109:39-69. doi: 10.1016/bs.adgen.2022.07.001. Epub 2022 Aug 18. Adv Genet. 2022. PMID: 36334916 Review.
Cited by
-
Characterization of the ArsRS regulon of Helicobacter pylori, involved in acid adaptation.J Bacteriol. 2006 May;188(10):3449-62. doi: 10.1128/JB.188.10.3449-3462.2006. J Bacteriol. 2006. PMID: 16672598 Free PMC article.
-
Insights into the Orchestration of Gene Transcription Regulators in Helicobacter pylori.Int J Mol Sci. 2022 Nov 8;23(22):13688. doi: 10.3390/ijms232213688. Int J Mol Sci. 2022. PMID: 36430169 Free PMC article. Review.
-
Holo-Ni(II)HpNikR is an asymmetric tetramer containing two different nickel-binding sites.J Am Chem Soc. 2010 Oct 20;132(41):14447-56. doi: 10.1021/ja104118r. J Am Chem Soc. 2010. PMID: 20863122 Free PMC article.
-
The nickel-responsive regulator NikR controls activation and repression of gene transcription in Helicobacter pylori.Infect Immun. 2005 Nov;73(11):7252-8. doi: 10.1128/IAI.73.11.7252-7258.2005. Infect Immun. 2005. PMID: 16239520 Free PMC article.
-
Iron-responsive regulation of the Helicobacter pylori iron-cofactored superoxide dismutase SodB is mediated by Fur.J Bacteriol. 2005 Jun;187(11):3687-92. doi: 10.1128/JB.187.11.3687-3692.2005. J Bacteriol. 2005. PMID: 15901691 Free PMC article.
References
-
- Akada, J. K., M. Shirai, H. Takeuchi, M. Tsuda, and T. Nakazawa. 2000. Identification of the urease operon in Helicobacter pylori and its control by mRNA decay in response to pH. Mol. Microbiol. 36:1071-1084. - PubMed
-
- Allan, E., C. L. Clayton, A. McLaren, D. M. Wallace, and B. W. Wren. 2001. Characterization of the low-pH responses of Helicobacter pylori using genomic DNA arrays. Microbiology 147:2285-2292. - PubMed
-
- Alm, R. A., L. S. Ling, D. T. Moir, B. L. King, E. D. Brown, P. C. Doig, D. R. Smith, B. Noonan, B. C. Guild, B. L. deJonge, G. Carmel, P. J. Tummino, A. Caruso, M. Uria-Nickelsen, D. M. Mills, C. Ives, R. Gibson, D. Merberg, S. D. Mills, Q. Jiang, D. E. Taylor, G. F. Vovis, and T. J. Trust. 1999. Genomic-sequence comparison of two unrelated isolates of the human gastric pathogen Helicobacter pylori. Nature 397:176-180. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

