Mutations in human complement regulator, membrane cofactor protein (CD46), predispose to development of familial hemolytic uremic syndrome
- PMID: 14566051
- PMCID: PMC240728
- DOI: 10.1073/pnas.2135497100
Mutations in human complement regulator, membrane cofactor protein (CD46), predispose to development of familial hemolytic uremic syndrome
Abstract
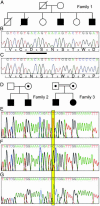
Membrane cofactor protein (MCP; CD46) is a widely expressed transmembrane complement regulator. Like factor H it inhibits complement activation by regulating C3b deposition on targets. Factor H mutations occur in 10-20% of patients with hemolytic uremic syndrome (HUS). We hypothesized that MCP mutations could predispose to HUS, and we sequenced MCP coding exons in affected individuals from 30 families. MCP mutations were detected in affected individuals of three families: a deletion of two amino acids (D237/S238) in family 1 (heterozygous) and a substitution, S206P, in families 2 (heterozygous) and 3 (homozygous). We evaluated protein expression and function in peripheral blood mononuclear cells from these individuals. An individual with the D237/S238 deletion had reduced MCP levels and approximately 50% C3b binding compared with normal controls. Individuals with the S206P change expressed normal quantities of protein, but demonstrated approximately 50% reduction in C3b binding in heterozygotes and complete lack of C3b binding in homozygotes. MCP expression and function was evaluated in transfectants reproducing these mutations. The deletion mutant was retained intracellularly. S206P protein was expressed on the cell surface but had a reduced ability to prevent complement activation, consistent with its reduced C3b binding and cofactor activity. This study presents further evidence that complement dysregulation predisposes to development of thrombotic microangiopathy and that screening patients for such defects could provide informed treatment strategies.
Figures

Similar articles
-
Modeling how CD46 deficiency predisposes to atypical hemolytic uremic syndrome.Mol Immunol. 2007 Mar;44(7):1559-68. doi: 10.1016/j.molimm.2006.08.024. Epub 2006 Oct 5. Mol Immunol. 2007. PMID: 17027083 Free PMC article.
-
Mutations in CD46, a complement regulatory protein, predispose to atypical HUS.Trends Mol Med. 2004 May;10(5):226-31. doi: 10.1016/j.molmed.2004.03.006. Trends Mol Med. 2004. PMID: 15121049 Review.
-
Familial haemolytic uraemic syndrome and an MCP mutation.Lancet. 2003 Nov 8;362(9395):1542-7. doi: 10.1016/S0140-6736(03)14742-3. Lancet. 2003. PMID: 14615110
-
Implications of the initial mutations in membrane cofactor protein (MCP; CD46) leading to atypical hemolytic uremic syndrome.Mol Immunol. 2007 Jan;44(1-3):111-22. doi: 10.1016/j.molimm.2006.07.004. Epub 2006 Aug 1. Mol Immunol. 2007. PMID: 16882452 Review.
-
Genetic and functional analyses of membrane cofactor protein (CD46) mutations in atypical hemolytic uremic syndrome.J Am Soc Nephrol. 2006 Jul;17(7):2017-25. doi: 10.1681/ASN.2005101051. Epub 2006 Jun 8. J Am Soc Nephrol. 2006. PMID: 16762990
Cited by
-
Complement disorders and hemolytic uremic syndrome.Curr Opin Pediatr. 2013 Apr;25(2):209-15. doi: 10.1097/MOP.0b013e32835df48a. Curr Opin Pediatr. 2013. PMID: 23399570 Free PMC article. Review.
-
Atypical hemolytic uremic syndrome in children: complement mutations and clinical characteristics.Pediatr Nephrol. 2012 Aug;27(8):1283-91. doi: 10.1007/s00467-012-2131-y. Epub 2012 Mar 13. Pediatr Nephrol. 2012. PMID: 22410797 Free PMC article.
-
Atypical Hemolytic Uremic Syndrome Triggered by Acute Pancreatitis in a Patient with a Membrane Cofactor Protein (CD46) Genetic Variant.Intern Med. 2024 Oct 1;63(19):2651-2654. doi: 10.2169/internalmedicine.3169-23. Epub 2024 Feb 26. Intern Med. 2024. PMID: 38403763 Free PMC article.
-
Shiga toxin 2 reduces complement inhibitor CD59 expression on human renal tubular epithelial and glomerular endothelial cells.Infect Immun. 2013 Aug;81(8):2678-85. doi: 10.1128/IAI.01079-12. Epub 2013 May 20. Infect Immun. 2013. PMID: 23690395 Free PMC article.
-
The glomerular filtration barrier: components and crosstalk.Int J Nephrol. 2012;2012:749010. doi: 10.1155/2012/749010. Epub 2012 Aug 14. Int J Nephrol. 2012. PMID: 22934182 Free PMC article.
References
-
- Moake, J. L. (2002) N. Engl. J. Med. 347, 589-600. - PubMed
-
- Richards, A., Goodship, J. A. & Goodship, T. H. J. (2002) Curr. Opin. Nephrol. Hypertens. 11, 431-435. - PubMed
-
- Warwicker, P., Goodship, T. H. J., Donne, R. L., Pirson, Y., Nicholls, A., Ward, R. M. & Goodship, J. A. (1998) Kidney Int. 53, 836-844. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Miscellaneous

