The tyrosyl-DNA phosphodiesterase Tdp1 is a member of the phospholipase D superfamily
- PMID: 11572945
- PMCID: PMC59758
- DOI: 10.1073/pnas.211429198
The tyrosyl-DNA phosphodiesterase Tdp1 is a member of the phospholipase D superfamily
Abstract
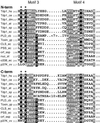
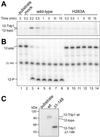
The phospholipase D (PLD) superfamily is a diverse group of proteins that includes enzymes involved in phospholipid metabolism, a bacterial toxin, poxvirus envelope proteins, and bacterial nucleases. Based on sequence comparisons, we show here that the tyrosyl-DNA phosphodiesterase (Tdp1) that has been implicated in the repair of topoisomerase I covalent complexes with DNA contains two unusual HKD signature motifs that place the enzyme in a distinct class within the PLD superfamily. Mutagenesis studies with the human enzyme in which the invariant histidines and lysines of the HKD motifs are changed confirm that these highly conserved residues are essential for Tdp1 activity. Furthermore, we show that, like other members of the family for which it has been examined, the reaction involves the formation of an intermediate in which the cleaved substrate is covalently linked to the enzyme. These results reveal that the hydrolytic reaction catalyzed by Tdp1 occurs by the phosphoryl transfer chemistry that is common to all members of the PLD superfamily.
Figures


Similar articles
-
The crystal structure of human tyrosyl-DNA phosphodiesterase, Tdp1.Structure. 2002 Feb;10(2):237-48. doi: 10.1016/s0969-2126(02)00707-4. Structure. 2002. PMID: 11839309
-
Structure-function studies of a plant tyrosyl-DNA phosphodiesterase provide novel insights into DNA repair mechanisms of Arabidopsis thaliana.Biochem J. 2012 Apr 1;443(1):49-56. doi: 10.1042/BJ20111308. Biochem J. 2012. PMID: 22214184 Free PMC article.
-
Analysis of the active-site mechanism of tyrosyl-DNA phosphodiesterase I: a member of the phospholipase D superfamily.J Mol Biol. 2012 Jan 27;415(4):741-58. doi: 10.1016/j.jmb.2011.11.044. Epub 2011 Dec 6. J Mol Biol. 2012. PMID: 22155078 Free PMC article.
-
Tyrosyl-DNA phosphodiesterase I resolves both naturally and chemically induced DNA adducts and its potential as a therapeutic target.Drug Metab Rev. 2014 Nov;46(4):494-507. doi: 10.3109/03602532.2014.971957. Epub 2014 Oct 20. Drug Metab Rev. 2014. PMID: 25327705 Review.
-
Tyrosyl-DNA Phosphodiesterase I N-Terminal Domain Modifications and Interactions Regulate Cellular Function.Genes (Basel). 2019 Nov 6;10(11):897. doi: 10.3390/genes10110897. Genes (Basel). 2019. PMID: 31698852 Free PMC article. Review.
Cited by
-
Critical roles of Rad54 in tolerance to apigenin-induced Top1-mediated DNA damage.Exp Ther Med. 2021 May;21(5):505. doi: 10.3892/etm.2021.9936. Epub 2021 Mar 18. Exp Ther Med. 2021. PMID: 33791014 Free PMC article.
-
Tyrosyl-DNA Phosphodiesterase I a critical survival factor for neuronal development and homeostasis.J Neurol Neuromedicine. 2016;1(5):25-29. doi: 10.29245/2572.942x/2016/5.1048. J Neurol Neuromedicine. 2016. PMID: 27747316 Free PMC article.
-
TDP1 phosphorylation by CDK1 in mitosis promotes MUS81-dependent repair of trapped Top1-DNA covalent complexes.EMBO J. 2024 Sep;43(17):3710-3732. doi: 10.1038/s44318-024-00169-3. Epub 2024 Jul 16. EMBO J. 2024. PMID: 39014228 Free PMC article.
-
Debulking of topoisomerase DNA-protein crosslinks (TOP-DPC) by the proteasome, non-proteasomal and non-proteolytic pathways.DNA Repair (Amst). 2020 Oct;94:102926. doi: 10.1016/j.dnarep.2020.102926. Epub 2020 Jul 10. DNA Repair (Amst). 2020. PMID: 32674013 Free PMC article. Review.
-
DNA repair functions that control sensitivity to topoisomerase-targeting drugs.Eukaryot Cell. 2004 Feb;3(1):82-90. doi: 10.1128/EC.3.1.82-90.2004. Eukaryot Cell. 2004. PMID: 14871939 Free PMC article.
References
-
- Morris A J, Engebrecht J, Frohman M A. Trends Pharmacol Sci. 1996;17:182–185. - PubMed
-
- Koonin E V. Trends Biochem Sci. 1996;21:242–243. - PubMed
-
- Stuckey J A, Dixon J E. Nat Struct Biol. 1999;6:278–284. - PubMed
-
- Leiros I, Secundo F, Zambonelli C, Servi S, Hough E. Struct Fold Des. 2000;8:655–667. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases

