Safety of Fezolinetant for Treatment of Moderate to Severe Vasomotor Symptoms Due to Menopause: Pooled Analysis of Three Randomized Phase 3 Studies
- PMID: 39739195
- PMCID: PMC11787274
- DOI: 10.1007/s12325-024-03073-8
Safety of Fezolinetant for Treatment of Moderate to Severe Vasomotor Symptoms Due to Menopause: Pooled Analysis of Three Randomized Phase 3 Studies
Abstract
Introduction: This study evaluated the safety and tolerability of fezolinetant in women with vasomotor symptoms (VMS) due to menopause in a pooled analysis of data from three 52-week phase 3 studies (SKYLIGHT 1, 2, and 4).
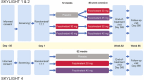
Methods: SKYLIGHT 1 and 2 were double-blind, placebo-controlled studies where women (≥ 40 to ≤ 65 years), with moderate to severe VMS (minimum average ≥ 7 hot flashes/day) were randomized to once-daily placebo, fezolinetant 30 mg or 45 mg. After 12 weeks, those on placebo were re-randomized to fezolinetant 30 mg or 45 mg, while those on fezolinetant continued on their assigned dose for 40 weeks. SKYLIGHT 4 was a placebo-controlled, double-blind, 52-week safety study. Safety was assessed by frequency of treatment-emergent adverse events (TEAEs) and endometrial events. TEAEs of special interest included liver test elevations and endometrial hyperplasia or cancer or disordered proliferative endometrium.
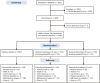
Results: Totals of 952 participants receiving placebo, 1100 receiving fezolinetant 45 mg, and 1103 receiving fezolinetant 30 mg took ≥ 1 dose of study medication. TEAEs occurred in 55.3%, 62.9%, and 65.4%, respectively; exposure-adjusted results were consistent with these results. Most frequent TEAEs in fezolinetant-treated participants included upper respiratory tract infection (7.7-8.3%), headache (6.8-8.2%), coronavirus disease 2019 (5.8-6.1%), back pain (3.1-3.7%), arthralgia (2.9-3.2%), diarrhea (2.3-3.2%), urinary tract infection (2.9-3.4%), and insomnia (2.0-3.0%). The incidence of drug-related serious TEAEs and associated treatment withdrawals was low. Elevations in liver transaminases occurred in 1.5-2.3% of fezolinetant-treated participants, were typically asymptomatic and transient, resolved on treatment or discontinuation, with no evidence of severe drug-induced liver injury (Hy's law). Endometrial safety results were well within US Food and Drug Administration criteria. Analysis of benign and non-benign neoplasm controlled for exposure demonstrated no increased risk versus placebo.
Conclusion: Pooled data confirm the safety and tolerability of fezolinetant over 52 weeks.
Trial registration: ClinicalTrials.gov identifiers, NCT04003155, NCT04003142, and NCT04003389. Graphical abstract available for this article.
Keywords: 52-week safety; Endometrial hyperplasia; Fezolinetant; Neurokinin 3 receptor antagonist; Transaminases; Treatment-emergent adverse events; Vasomotor symptoms.
© 2024. The Author(s).
Conflict of interest statement
Declarations. Conflict of Interest: RK has received consulting/advisory board fees from Astellas, Pfizer, and Mayne, participated in the data monitoring committee for Astellas (ended), received speaker fees from Pfizer and Astellas, and acted as Medical Advisor for Carrot Fertility. AC acted as former President of the European Menopause and Andropause Society and consulted for Astellas, Theramex, and Viatris. REN received grants from Fidia, consulting fees from Astellas, Besins, Bayer, Exeltis, Fidia, Freya, HRA, Theramex, and Vichy Laboratories, honoraria for lectures from Abbott, Bayer, Exeltis, Novo Nordisk, Organon, Shionogi, Theramex, and Viatris, was a researcher for Fidia, Merck, and Shionogi, and is President Elect of the International Menopause Society. MLE, SM, XW, and FDO are employees of Astellas. Ethical Approval: SKYLIGHT 1, 2, and 4 were conducted in accordance with Declaration of Helsinki, Good Clinical Practice, and International Council for Harmonisation guidelines. An independent ethics committee or institutional review board reviewed ethical, scientific, and medical appropriateness of the study at each site before data collection. Written informed consent was obtained from all participants before any study-related procedures.
Figures
Similar articles
-
Efficacy and safety of fezolinetant for moderate-severe vasomotor symptoms associated with menopause in individuals unsuitable for hormone therapy: phase 3b randomised controlled trial.BMJ. 2024 Nov 18;387:e079525. doi: 10.1136/bmj-2024-079525. BMJ. 2024. PMID: 39557487 Free PMC article. Clinical Trial.
-
Phase II study of fezolinetant for treatment of vasomotor symptoms associated with menopause in Japan.Climacteric. 2024 Aug;27(4):389-397. doi: 10.1080/13697137.2024.2356854. Epub 2024 Jun 12. Climacteric. 2024. PMID: 38864290 Clinical Trial.
-
Effectiveness and safety of fezolinetant in alleviating vasomotor symptoms linked to Menopause.: A systematic review and Meta-Analysis.Eur J Obstet Gynecol Reprod Biol. 2024 Jun;297:142-152. doi: 10.1016/j.ejogrb.2024.04.017. Epub 2024 Apr 15. Eur J Obstet Gynecol Reprod Biol. 2024. PMID: 38640780
-
Efficacy and safety of fezolinetant for vasomotor symptoms in postmenopausal women: A systematic review and meta-analysis of randomized controlled trials.Int J Gynaecol Obstet. 2024 Sep;166(3):969-983. doi: 10.1002/ijgo.15467. Epub 2024 Apr 2. Int J Gynaecol Obstet. 2024. PMID: 38563867
-
Oral budesonide for induction of remission in ulcerative colitis.Cochrane Database Syst Rev. 2015 Oct 26;2015(10):CD007698. doi: 10.1002/14651858.CD007698.pub3. Cochrane Database Syst Rev. 2015. PMID: 26497719 Free PMC article. Review.
References
-
- Monteleone P, Mascagni G, Giannini A, Genazzani AR, Simoncini T. Symptoms of menopause - global prevalence, physiology and implications. Nat Rev Endocrinol. 2018;14(4):199–215. - PubMed
-
- Voedisch AJ, Dunsmoor-Su R, Kasirsky J. Menopause: a global perspective and clinical guide for practice. Clin Obstet Gynecol. 2021;64(3):528–54. - PubMed
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Medical

