Reduced hepatic impairment study to evaluate pharmacokinetics and safety of zavegepant and to inform dosing recommendation for hepatic impairment
- PMID: 39014555
- PMCID: PMC11252018
- DOI: 10.1111/cts.13813
Reduced hepatic impairment study to evaluate pharmacokinetics and safety of zavegepant and to inform dosing recommendation for hepatic impairment
Abstract
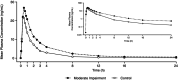
Zavegepant, a high-affinity, selective, small-molecule calcitonin gene-related peptide (CGRP) receptor antagonist, is approved in the United States for acute treatment of migraine in adults. The effects of moderate hepatic impairment (8 participants with Child-Pugh score 7-9 points) on the pharmacokinetics of a single 10-mg intranasal dose of zavegepant versus eight matched participants with normal hepatic function were evaluated in a phase I study. Pharmacokinetic sampling determined total and unbound plasma zavegepant concentrations. Moderate hepatic impairment increased the exposure of total zavegepant (~2-fold increase in AUC0-inf and 16% increase in Cmax) versus normal hepatic function, which is not considered clinically meaningful. The geometric least squares mean ratios (moderate impairment/normal) of plasma zavegepant AUC0-inf and Cmax were 193% (90% confidence interval [CI]: 112, 333; p = 0.051) and 116% (90% CI: 69, 195; p = 0.630), respectively. The geometric mean fraction unbound of zavegepant was similar for participants with moderate hepatic impairment (0.13; coefficient of variation [CV] 13.71%) versus those with normal hepatic function (0.11; CV 21.43%). Similar exposure findings were observed with unbound zavegepant versus normal hepatic function (~2.3-fold increase in AUC0-inf and 39% increase in Cmax). One treatment-emergent adverse event (mild, treatment-related headache) was reported in a participant with normal hepatic function. No dosage adjustment of intranasal zavegepant is required in adults with mild or moderate hepatic impairment.
© 2024 Pfizer Inc and The Authors. Clinical and Translational Science published by Wiley Periodicals LLC on behalf of American Society for Clinical Pharmacology and Therapeutics.
Conflict of interest statement
R.B. is an employee of Certara who was a paid consultant to Biohaven, acquired by Pfizer in October 2022, for this study. M.K.D., J.M., B.M., R.B. are current or former employees of Biohaven, acquired by Pfizer in October 2022, for this study. R.C. was an employee of Biohaven Pharmaceuticals, owns stock in Biohaven Ltd, was an employee of Pfizer, has received research payments from Pfizer, and provides services to Collima LLC, which has had consulting agreements with Pfizer, Aptose Biosciences Inc., Manistee Therapeutics, and Vida Ventures Management Co., LLC. T.C.M. is an employee and equity owner of Orlando Clinical Research Center. K.T.M., J.L. are employed by and hold stock/options in Pfizer.
Figures

Similar articles
-
Long-term safety of zavegepant nasal spray for the acute treatment of migraine: A phase 2/3 open-label study.Cephalalgia. 2024 Aug;44(8):3331024241259456. doi: 10.1177/03331024241259456. Cephalalgia. 2024. PMID: 39210835 Clinical Trial.
-
Comparative bioavailability of single-dose zavegepant during and between migraine attacks: A phase 1, randomized, open-label, fixed-sequence, two-period study.Headache. 2025 Mar;65(3):484-494. doi: 10.1111/head.14856. Epub 2024 Nov 7. Headache. 2025. PMID: 39508426 Free PMC article. Clinical Trial.
-
Comparison of gepant effects at therapeutic plasma concentrations: connecting pharmacodynamics and pharmacokinetics.J Headache Pain. 2024 Aug 28;25(1):141. doi: 10.1186/s10194-024-01846-8. J Headache Pain. 2024. PMID: 39198753 Free PMC article.
-
Zavegepant Intranasal Spray for Migraines.Ann Pharmacother. 2024 Aug;58(8):827-833. doi: 10.1177/10600280231209439. Epub 2023 Oct 28. Ann Pharmacother. 2024. PMID: 37897226 Review.
-
Focus on zavegepant: the first intranasal third-generation gepant.Pain Manag. 2022 Nov;12(8):879-885. doi: 10.2217/pmt-2022-0054. Epub 2022 Oct 3. Pain Manag. 2022. PMID: 36189708 Review.
References
-
- International Headache Society . Headache classification Committee of the International Headache Society (IHS) the international classification of headache disorders, 3rd edition. Cephalalgia. 2018;38:1‐211. - PubMed
-
- Burch R, Rizzoli P, Loder E. The prevalence and impact of migraine and severe headache in the United States: figures and trends from government health studies. Headache. 2018;58:496‐505. - PubMed
-
- World Health Organization . Headache disorders. 2016. Accessed March 22, 2022. www.who.int/news‐room/fact‐sheets/detail/headache‐disorders
-
- GBD 2016 Disease and Injury Incidence and Prevalence Collaborators . Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: a systematic analysis for the global burden of disease study 2016. Lancet. 2017;390:1211‐1259. - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

