Decreased pyruvate dehydrogenase activity in Tafazzin-deficient cells is caused by dysregulation of pyruvate dehydrogenase phosphatase 1 (PDP1)
- PMID: 38301889
- PMCID: PMC10884759
- DOI: 10.1016/j.jbc.2024.105697
Decreased pyruvate dehydrogenase activity in Tafazzin-deficient cells is caused by dysregulation of pyruvate dehydrogenase phosphatase 1 (PDP1)
Abstract
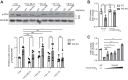
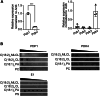
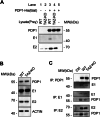
Cardiolipin (CL), the signature lipid of the mitochondrial inner membrane, is critical for maintaining optimal mitochondrial function and bioenergetics. Disruption of CL metabolism, caused by mutations in the CL remodeling enzyme TAFAZZIN, results in the life-threatening disorder Barth syndrome (BTHS). While the clinical manifestations of BTHS, such as dilated cardiomyopathy and skeletal myopathy, point to defects in mitochondrial bioenergetics, the disorder is also characterized by broad metabolic dysregulation, including abnormal levels of metabolites associated with the tricarboxylic acid (TCA) cycle. Recent studies have identified the inhibition of pyruvate dehydrogenase (PDH), the gatekeeper enzyme for TCA cycle carbon influx, as a key deficiency in various BTHS model systems. However, the molecular mechanisms linking aberrant CL remodeling, particularly the primary, direct consequence of reduced tetralinoleoyl-CL (TLCL) levels, to PDH activity deficiency are not yet understood. In the current study, we found that remodeled TLCL promotes PDH function by directly binding to and enhancing the activity of PDH phosphatase 1 (PDP1). This is supported by our findings that TLCL uniquely activates PDH in a dose-dependent manner, TLCL binds to PDP1 in vitro, TLCL-mediated PDH activation is attenuated in the presence of phosphatase inhibitor, and PDP1 activity is decreased in Tafazzin-knockout (TAZ-KO) C2C12 myoblasts. Additionally, we observed decreased mitochondrial calcium levels in TAZ-KO cells and treating TAZ-KO cells with calcium lactate (CaLac) increases mitochondrial calcium and restores PDH activity and mitochondrial oxygen consumption rate. Based on our findings, we conclude that reduced mitochondrial calcium levels and decreased binding of PDP1 to TLCL contribute to decreased PDP1 activity in TAZ-KO cells.
Keywords: Barth syndrome; calcium; caridiolipin; metabolism; pyruvate dehdrogenase phosphatase; pyruvate dehydrogenase; tafazzin.
Copyright © 2024 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Conflict of interest The authors declare that they have no conflicts of interest with the contents of this article.
Figures
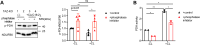


Similar articles
-
Stem cell models of TAFAZZIN deficiency reveal novel tissue-specific pathologies in Barth syndrome.Hum Mol Genet. 2025 Jan 23;34(1):101-115. doi: 10.1093/hmg/ddae152. Hum Mol Genet. 2025. PMID: 39535077
-
The lipid environment modulates cardiolipin and phospholipid constitution in wild type and tafazzin-deficient cells.J Inherit Metab Dis. 2022 Jan;45(1):38-50. doi: 10.1002/jimd.12433. Epub 2021 Sep 28. J Inherit Metab Dis. 2022. PMID: 34494285
-
Upregulation of the AMPK-FOXO1-PDK4 pathway is a primary mechanism of pyruvate dehydrogenase activity reduction and leads to increased glucose uptake in tafazzin-deficient cells.bioRxiv [Preprint]. 2024 Feb 4:2024.02.03.578755. doi: 10.1101/2024.02.03.578755. bioRxiv. 2024. Update in: Sci Rep. 2024 May 20;14(1):11497. doi: 10.1038/s41598-024-62262-1. PMID: 38352304 Free PMC article. Updated. Preprint.
-
Barth Syndrome.2014 Oct 9 [updated 2020 Jul 9]. In: Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2025. 2014 Oct 9 [updated 2020 Jul 9]. In: Adam MP, Feldman J, Mirzaa GM, Pagon RA, Wallace SE, Amemiya A, editors. GeneReviews® [Internet]. Seattle (WA): University of Washington, Seattle; 1993–2025. PMID: 25299040 Free Books & Documents. Review.
-
Barth syndrome.Am J Med Genet C Semin Med Genet. 2013 Aug;163C(3):198-205. doi: 10.1002/ajmg.c.31372. Epub 2013 Jul 10. Am J Med Genet C Semin Med Genet. 2013. PMID: 23843353 Free PMC article. Review.
Cited by
-
Upregulation of the AMPK-FOXO1-PDK4 pathway is a primary mechanism of pyruvate dehydrogenase activity reduction in tafazzin-deficient cells.Sci Rep. 2024 May 20;14(1):11497. doi: 10.1038/s41598-024-62262-1. Sci Rep. 2024. PMID: 38769106 Free PMC article.
-
The Role of Cardiolipin in Brain Bioenergetics, Neuroinflammation, and Neurodegeneration.Mol Neurobiol. 2024 Nov 19. doi: 10.1007/s12035-024-04630-6. Online ahead of print. Mol Neurobiol. 2024. PMID: 39557801 Review.
References
-
- Barth P.G., Wanders R.J., Vreken P., Janssen E.A., Lam J., Baas F. X-linked cardioskeletal myopathy and neutropenia (Barth syndrome) (MIM 302060) J. Inherit. Metab. Dis. 1999;22:555–567. - PubMed
-
- Xu Y., Kelley R.I., Blanck T.J.J., Schlame M. Remodeling of cardiolipin by phospholipid transacylation. J. Biol. Chem. 2003;278:51380–51385. - PubMed
-
- Schlame M., Ren M. Barth syndrome, a human disorder of cardiolipin metabolism. FEBS Lett. 2006;580:5450–5455. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

