Clinically Important Benefits and Harms of Monoclonal Antibodies Targeting Amyloid for the Treatment of Alzheimer Disease: A Systematic Review and Meta-Analysis
- PMID: 38253509
- PMCID: PMC11233076
- DOI: 10.1370/afm.3050
Clinically Important Benefits and Harms of Monoclonal Antibodies Targeting Amyloid for the Treatment of Alzheimer Disease: A Systematic Review and Meta-Analysis
Abstract
Purpose: We conducted a meta-analysis to evaluate clinically meaningful benefits and harms of monoclonal antibodies targeting amyloid in patients with Alzheimer dementia.
Methods: We searched PubMed, Cochrane CENTRAL, and 5 trial registries, as well as the reference lists of identified studies. We included randomized controlled trials comparing a monoclonal antibody with placebo at a dose consistent with that used in phase 3 trials or for Food and Drug Administration approval. Studies had to report at least 1 clinically relevant benefit or harm. Data were extracted independently by at least 2 researchers for random effects meta-analysis. Changes in cognitive and functional scales were compared between groups, and each difference was assessed to determine if it met the minimal clinically important difference (MCID).
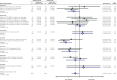
Results: We identified 19 publications with 23,202 total participants that evaluated 8 anti-amyloid antibodies. There were small improvements over placebo in the Alzheimer's Disease Assessment Scale (ADAS)-Cog-11 to -14 score (standardized mean difference = -0.07; 95% CI, -0.10 to -0.04), Mini Mental State Examination score (0.32 points; 95% CI, 0.13 to 0.50), and Clinical Dementia Rating-Sum of Boxes scale score (mean difference =-0.18 points; 95% CI, -0.34 to -0.03), and the combined functional scores (standardized mean difference = 0.09; 95% CI, 0.05 to 0.13). None of the changes, including those for lecanemab, aducanumab, and donanemab, exceeded the MCID. Harms included significantly increased risks of amyloid-related imaging abnormalities (ARIA)-edema (relative risk [RR] = 10.29; number needed to harm [NNH] = 9), ARIA-hemorrhage (RR = 1.74; NNH = 13), and symptomatic ARIA-edema (RR = 24.3; NNH = 86).
Conclusions: Although monoclonal antibodies targeting amyloid provide small benefits on cognitive and functional scales in patients with Alzheimer dementia, these improvements are far below the MCID for each outcome and are accompanied by clinically meaningful harms.
Keywords: ARIA; Alzheimer dementia; Alzheimer disease; aducanumab; aged; amyloid; antibodies, monoclonal; biological therapy; cerebral edema; cerebral hemorrhage; chronic disease; dementia; donanemab; drug approval; lecanemab; meta-analysis; risks and benefits; systematic review.
© 2024 Annals of Family Medicine, Inc.
Figures



Similar articles
-
Comparative Efficacy and Safety of Monoclonal Antibodies for Cognitive Decline in Patients with Alzheimer's Disease: A Systematic Review and Network Meta-Analysis.CNS Drugs. 2024 Mar;38(3):169-192. doi: 10.1007/s40263-024-01067-2. Epub 2024 Mar 1. CNS Drugs. 2024. PMID: 38429615
-
Efficacy of anti-amyloid-ß monoclonal antibody therapy in early Alzheimer's disease: a systematic review and meta-analysis.Neurol Sci. 2024 Jun;45(6):2461-2469. doi: 10.1007/s10072-023-07194-w. Epub 2023 Nov 17. Neurol Sci. 2024. PMID: 37978096
-
Comparative efficacy, tolerability and acceptability of donanemab, lecanemab, aducanumab and lithium on cognitive function in mild cognitive impairment and Alzheimer's disease: A systematic review and network meta-analysis.Ageing Res Rev. 2024 Feb;94:102203. doi: 10.1016/j.arr.2024.102203. Epub 2024 Jan 20. Ageing Res Rev. 2024. PMID: 38253184 Review.
-
The FDA-approved anti-amyloid-β monoclonal antibodies for the treatment of Alzheimer's disease: a systematic review and meta-analysis of randomized controlled trials.Eur J Med Res. 2023 Nov 28;28(1):544. doi: 10.1186/s40001-023-01512-w. Eur J Med Res. 2023. PMID: 38017568 Free PMC article. Review.
-
Amyloid-Related Imaging Abnormalities in 2 Phase 3 Studies Evaluating Aducanumab in Patients With Early Alzheimer Disease.JAMA Neurol. 2022 Jan 1;79(1):13-21. doi: 10.1001/jamaneurol.2021.4161. JAMA Neurol. 2022. PMID: 34807243 Free PMC article. Clinical Trial.
Cited by
-
Alzheimer's Amyloid Hypothesis and Antibody Therapy: Melting Glaciers?Int J Mol Sci. 2024 Mar 31;25(7):3892. doi: 10.3390/ijms25073892. Int J Mol Sci. 2024. PMID: 38612701 Free PMC article. Review.
-
Exploring Tau Fibril-Disaggregating and Antioxidating Molecules Binding to Membrane-Bound Amyloid Oligomers Using Machine Learning-Enhanced Docking and Molecular Dynamics.Molecules. 2024 Jun 13;29(12):2818. doi: 10.3390/molecules29122818. Molecules. 2024. PMID: 38930883 Free PMC article.
-
Monoamine alterations in Alzheimer's disease and their implications in comorbid neuropsychiatric symptoms.Geroscience. 2025 Feb;47(1):457-482. doi: 10.1007/s11357-024-01359-x. Epub 2024 Sep 27. Geroscience. 2025. PMID: 39331291 Free PMC article. Review.
-
Progressive Age-Associated Blood-Brain Barrier Leak/Dysfunction-Nexus of Neurodegenerative Disease Using MRI Markers to Identify Preclinical Disease and Potential New Targets for Future Treatments.Diagnostics (Basel). 2024 Mar 29;14(7):726. doi: 10.3390/diagnostics14070726. Diagnostics (Basel). 2024. PMID: 38611639 Free PMC article. Review.
-
Hippocampal CA1 Pyramidal Neurons Display Sublayer and Circuitry Dependent Degenerative Expression Profiles in Aged Female Down Syndrome Mice.J Alzheimers Dis. 2024;100(s1):S341-S362. doi: 10.3233/JAD-240622. J Alzheimers Dis. 2024. PMID: 39031371 Free PMC article.
References
-
- US Food and Drug Administration . FDA’s decision to approve new treatment for Alzheimer’s disease. Published 2021. Accessed Dec 14, 2023. https://www.fda.gov/drugs/our-perspective/fdas-decision-approve-new-trea...
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
