Impact of CRAMP-34 on Pseudomonas aeruginosa biofilms and extracellular metabolites
- PMID: 38162583
- PMCID: PMC10757720
- DOI: 10.3389/fcimb.2023.1295311
Impact of CRAMP-34 on Pseudomonas aeruginosa biofilms and extracellular metabolites
Abstract
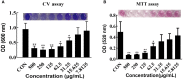
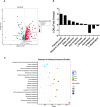
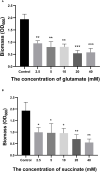
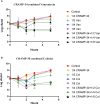
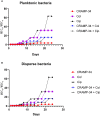
Biofilm is a structured community of bacteria encased within a self-produced extracellular matrix. When bacteria form biofilms, they undergo a phenotypic shift that enhances their resistance to antimicrobial agents. Consequently, inducing the transition of biofilm bacteria to the planktonic state may offer a viable approach for addressing infections associated with biofilms. Our previous study has shown that the mouse antimicrobial peptide CRAMP-34 can disperse Pseudomonas aeruginosa (P. aeruginosa) biofilm, and the potential mechanism of CRAMP-34 eradicate P. aeruginosa biofilms was also investigated by combined omics. However, changes in bacterial extracellular metabolism have not been identified. To further explore the mechanism by which CRAMP-34 disperses biofilm, this study analyzed its effects on the extracellular metabolites of biofilm cells via metabolomics. The results demonstrated that a total of 258 significantly different metabolites were detected in the untargeted metabolomics, of which 73 were downregulated and 185 were upregulated. Pathway enrichment analysis of differential metabolites revealed that metabolic pathways are mainly related to the biosynthesis and metabolism of amino acids, and it also suggested that CRAMP-34 may alter the sensitivity of biofilm bacteria to antibiotics. Subsequently, it was confirmed that the combination of CRAMP-34 with vancomycin and colistin had a synergistic effect on dispersed cells. These results, along with our previous findings, suggest that CRAMP-34 may promote the transition of PAO1 bacteria from the biofilm state to the planktonic state by upregulating the extracellular glutamate and succinate metabolism and eventually leading to the dispersal of biofilm. In addition, increased extracellular metabolites of myoinositol, palmitic acid and oleic acid may enhance the susceptibility of the dispersed bacteria to the antibiotics colistin and vancomycin. CRAMP-34 also delayed the development of bacterial resistance to colistin and ciprofloxacin. These results suggest the promising development of CRAMP-34 in combination with antibiotics as a potential candidate to provide a novel therapeutic approach for the prevention and treatment of biofilm-associated infections.
Keywords: CRAMP-34; Pseudomonas aeruginosa; anti-biofilm peptides; extracellular metabolites; synergistic effect.
Copyright © 2023 Wang, Ma, Long, Cheng, Zhang, Peng, Fu, Yu, Xu, Zhang, Qiu, He, Yang and Chen.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Pseudomonas aeruginosa biofilm dispersion by the mouse antimicrobial peptide CRAMP.Vet Res. 2022 Oct 8;53(1):80. doi: 10.1186/s13567-022-01097-y. Vet Res. 2022. PMID: 36209206 Free PMC article.
-
Effects of a novel anti-biofilm peptide CRAMP combined with antibiotics on the formation of Pseudomonas aeruginosa biofilms.Microb Pathog. 2021 Mar;152:104660. doi: 10.1016/j.micpath.2020.104660. Epub 2020 Nov 27. Microb Pathog. 2021. PMID: 33253855
-
D-amino acids enhance the activity of antimicrobials against biofilms of clinical wound isolates of Staphylococcus aureus and Pseudomonas aeruginosa.Antimicrob Agents Chemother. 2014 Aug;58(8):4353-61. doi: 10.1128/AAC.02468-14. Epub 2014 May 19. Antimicrob Agents Chemother. 2014. PMID: 24841260 Free PMC article.
-
Pseudomonas aeruginosa biofilm infections: from molecular biofilm biology to new treatment possibilities.APMIS Suppl. 2014 Dec;(138):1-51. doi: 10.1111/apm.12335. APMIS Suppl. 2014. PMID: 25399808 Review.
-
Standard versus biofilm antimicrobial susceptibility testing to guide antibiotic therapy in cystic fibrosis.Cochrane Database Syst Rev. 2017 Oct 5;10(10):CD009528. doi: 10.1002/14651858.CD009528.pub4. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2020 Jun 10;6:CD009528. doi: 10.1002/14651858.CD009528.pub5. PMID: 28981972 Free PMC article. Updated. Review.
Cited by
-
Effect of anti-biofilm peptide CRAMP-34 on the biofilms of Acinetobacter lwoffii derived from dairy cows.Front Cell Infect Microbiol. 2024 Aug 15;14:1406429. doi: 10.3389/fcimb.2024.1406429. eCollection 2024. Front Cell Infect Microbiol. 2024. PMID: 39211795 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

