The Comparison between Vadadustat and Daprodustat Regarding Dose, Cost, and Safety of Treatment for Renal Anemia in Non-dialysis Patients with Chronic Kidney Diseases
- PMID: 37926547
- PMCID: PMC11272495
- DOI: 10.2169/internalmedicine.2501-23
The Comparison between Vadadustat and Daprodustat Regarding Dose, Cost, and Safety of Treatment for Renal Anemia in Non-dialysis Patients with Chronic Kidney Diseases
Abstract
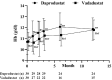
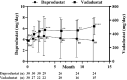
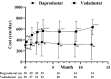
Objective We retrospectively compared the dose, cost, and safety of vadadustat and daprodustat for the treatment of renal anemia in patients with chronic kidney diseases who were not undergoing dialysis. Methods The primary outcome of this study was the change in dose and cost from the initiation of vadadustat and daprodustat treatment. The secondary outcome was the drug safety. Patients We treated 30 patients each with the hypoxia-inducible factor prolyl-hydroxylase inhibitors (HIF-PHIs) daprodustat and vadadustat. The hemoglobin (Hb) concentration was targeted at 11-13 g/dL, and transferrin saturation was maintained at ≥20%, as per the 2018 Japanese guidelines for the diagnosis and treatment of chronic kidney disease. Results Hb levels increased from 10.7 to 11.5 g/dL after the first month of daprodustat administration, whereas those for vadadustat patients remained relatively stable, going from 10.7 to 10.6 g/dL. After six months, the Hb level reached 12.1 g/dL and 11.3 g/dL for daprodustat and vadadustat, respectively. The dosage of vadadustat was significantly increased by 46% and 70% after 3 and 12 months, respectively, compared with the initial doses, whereas that of daprodustat did not change substantially. The average cost of vadadustat also increased in the first 3 months and remained over 500 yen/day after 3 months, while that of daprodustat showed little change from the initial cost of 360 yen/day. Conclusion These results suggest that heterogeneity exists in the drug potency and dosage required for treatment between daprodustat and vadadustat. Serious adverse events [death, cardiovascular disease, end stage renal disease (ESRD), and malignancy] occurred in more than 20% of participants with both HIF-PHIs. Further studies are required to confirm the safety of HIF-PHIs.
Keywords: HIF-PH inhibitor; cost; dose; renal anemia; safety.
Conflict of interest statement
Figures

Similar articles
-
The comparison of four hypoxia-inducible factor prolyl hydroxylase inhibitors on drug potency and cost for treatment in patients with renal anemia.Clin Exp Nephrol. 2024 Nov;28(11):1090-1096. doi: 10.1007/s10157-024-02511-9. Epub 2024 May 20. Clin Exp Nephrol. 2024. PMID: 38767688
-
Daprodustat: A Hypoxia-Inducible Factor-Prolyl Hydroxylase Inhibitor for Anemia of Chronic Kidney Disease.Ann Pharmacother. 2025 Jan;59(1):71-80. doi: 10.1177/10600280241241563. Epub 2024 Apr 14. Ann Pharmacother. 2025. PMID: 38616529 Review.
-
A randomized, 29-day, dose-ranging, efficacy and safety study of daprodustat, administered three times weekly in patients with anemia on hemodialysis.BMC Nephrol. 2019 Oct 16;20(1):372. doi: 10.1186/s12882-019-1547-z. BMC Nephrol. 2019. PMID: 31619187 Free PMC article. Clinical Trial.
-
Efficacy and safety of HIF prolyl-hydroxylase inhibitor vs epoetin and darbepoetin for anemia in chronic kidney disease patients not undergoing dialysis: A network meta-analysis.Pharmacol Res. 2020 Sep;159:105020. doi: 10.1016/j.phrs.2020.105020. Epub 2020 Jun 16. Pharmacol Res. 2020. PMID: 32561478 Review.
-
Effects of Daprodustat, a Novel Hypoxia-Inducible Factor Prolyl Hydroxylase Inhibitor on Anemia Management in Japanese Hemodialysis Subjects.Am J Nephrol. 2017;45(2):127-135. doi: 10.1159/000454818. Epub 2016 Dec 16. Am J Nephrol. 2017. PMID: 27978511 Clinical Trial.
Cited by
-
Chemistry, Analysis, and Biological Aspects of Daprodustat, A New Hypoxia Inducible Factor Prolyl Hydroxylase Inhibitor: A Comprehensive Review.Mini Rev Med Chem. 2024;24(20):1847-1855. doi: 10.2174/0113895575293447240424052516. Mini Rev Med Chem. 2024. PMID: 38685804 Review.
References
-
- Silverberg DS, Wexler D, Iaina A, Steinbruch S, Wollman Y, Schwartz D. Anemia, chronic renal disease and congestive heart failure - the cardio renal anemia syndrome: the need for cooperation between cardiologists and nephrologists. Int Urol Nephrol 38: 295-310, 2006. - PubMed
-
- Macdougall IC, Lewis NP, Saunders MJ, et al. . Long-term cardiorespiratory effects of amelioration of renal anaemia by erythropoietin. Lancet 335: 489-493, 1990. - PubMed
-
- Sugahara M, Tanaka T, Nangaku M. Future perspectives of anemia management in chronic kidney disease using hypoxia-inducible factor-prolyl hydroxylase inhibitors. Pharmacol Ther 239: 108272, 2022. - PubMed

