Interim analysis: Open-label extension study of leniolisib for patients with APDS
- PMID: 37797893
- PMCID: PMC10841669
- DOI: 10.1016/j.jaci.2023.09.032
Interim analysis: Open-label extension study of leniolisib for patients with APDS
Abstract
Background: Activated phosphoinositide 3-kinase delta (PI3Kδ) syndrome (APDS; or p110δ-activating mutations causing senescent T cells, lymphadenopathy, and immunodeficiency) is an inborn error of immunity caused by PI3Kδ hyperactivity. Resultant immune deficiency and dysregulation lead to recurrent sinopulmonary infections, herpes viremia, autoimmunity, and lymphoproliferation.
Objective: Leniolisib, a selective PI3Kδ inhibitor, demonstrated favorable impact on immune cell subsets and lymphoproliferation over placebo in patients with APDS over 12 weeks. Here, we report results from an interim analysis of an ongoing open-label, single-arm extension study.
Methods: Patients with APDS aged 12 years or older who completed NCT02435173 or had previous exposure to PI3Kδ inhibitors were eligible. The primary end point was safety, assessed via investigator-reported adverse events (AEs) and clinical/laboratory evaluations. Secondary and exploratory end points included health-related quality of life, inflammatory markers, frequency of infections, and lymphoproliferation.
Results: Between September 2016 and August 2021, 37 patients (median age, 20 years; 42.3% female) were enrolled. Of these 37 patients, 26, 9, and 2 patients had previously received leniolisib, placebo, or other PI3Kδ inhibitors, respectively. At the data cutoff date (December 13, 2021), median leniolisib exposure was 102 weeks. Overall, 32 patients (87%) experienced an AE. Most AEs were grades 1 to 3; none were grade 4. One patient with severe baseline comorbidities experienced a grade 5 AE, determined as unrelated to leniolisib treatment. While on leniolisib, patients had reduced annualized infection rates (P = .004), and reductions in immunoglobulin replacement therapy occurred in 10 of 27 patients. Other observations include reduced lymphadenopathy and splenomegaly, improved cytopenias, and normalized lymphocyte subsets.
Conclusions: Leniolisib was well tolerated and maintained durable outcomes with up to 5 years of exposure in 37 patients with APDS.
Clinicaltrials: gov identifier: NCT02859727.
Keywords: APDS; B cells; PI3Kδ inhibitor; PIK3CD; PIK3R1; T cells; clinical trial; long-term safety; lymphoproliferation; primary immunodeficiency.
Published by Elsevier Inc.
Conflict of interest statement
Conflict Of Interest Disclosures
V.K.R., A.P., V.L., A.O., G.U., S.M.J., and M.J.L. have no competing financial interests. S.W. is a consultant for Pharming Group, NV. A. Šedivá is a consultant for Octapharma, Takeda, and Pharming NV. A. Shcherbina receives honoraria from and is a consultant for Octapharma, CSL Behring, and Novartis. E.K. is an employee of Leidos Biomedical Research, Inc. J.K. has received honorarium from Pharming Group, NV. V.A.D. is a consultant for and/or receives honoraria from AstraZeneca, Kedrion, Takeda, CSL Behring, Pfizer, and Pharming Group, NV. V.A.D. also receives honoraria from Pfizer, and research funding from Takeda. K.K. is an employee and shareholder of Novartis Pharma AG. K.R. is an employee of Novartis Pharma AG. A.R. and J.B. are current employees and stock option holders of Pharming Group, NV, and J.B. holds individual stock in Neoclone.
Figures


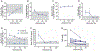
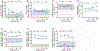

Similar articles
-
A randomised, placebo-controlled, phase III trial of leniolisib in activated phosphoinositide 3-kinase delta (PI3Kδ) syndrome (APDS): Adolescent and adult subgroup analysis.Clin Immunol. 2025 Jan;270:110400. doi: 10.1016/j.clim.2024.110400. Epub 2024 Nov 17. Clin Immunol. 2025. PMID: 39561927 Clinical Trial.
-
Report of the Italian Cohort with Activated Phosphoinositide 3-Kinase δ Syndrome in the Target Therapy Era.J Clin Immunol. 2024 Dec 23;45(1):58. doi: 10.1007/s10875-024-01835-1. J Clin Immunol. 2024. PMID: 39714594 Free PMC article.
-
Comparative efficacy of leniolisib (CDZ173) versus standard of care on rates of respiratory tract infection and serum immunoglobulin M (IgM) levels among individuals with activated phosphoinositide 3-kinase delta (PI3Kδ) syndrome (APDS): an externally controlled study.Clin Exp Immunol. 2025 Jan 21;219(1):uxae107. doi: 10.1093/cei/uxae107. Clin Exp Immunol. 2025. PMID: 39673396 Free PMC article. Clinical Trial.
-
Azathioprine and 6-mercaptopurine for maintenance of surgically-induced remission in Crohn's disease.Cochrane Database Syst Rev. 2014 Aug 1;2014(8):CD010233. doi: 10.1002/14651858.CD010233.pub2. Cochrane Database Syst Rev. 2014. Update in: Cochrane Database Syst Rev. 2019 Aug 06;8:CD010233. doi: 10.1002/14651858.CD010233.pub3. PMID: 25081347 Free PMC article. Updated. Review.
-
Oral budesonide for induction of remission in ulcerative colitis.Cochrane Database Syst Rev. 2015 Oct 26;2015(10):CD007698. doi: 10.1002/14651858.CD007698.pub3. Cochrane Database Syst Rev. 2015. PMID: 26497719 Free PMC article. Review.
Cited by
-
Monogenic Inborn Errors of Immunity with impaired IgG response to polysaccharide antigens but normal IgG levels and normal IgG response to protein antigens.Front Pediatr. 2024 Jun 12;12:1386959. doi: 10.3389/fped.2024.1386959. eCollection 2024. Front Pediatr. 2024. PMID: 38933494 Free PMC article. Review.
-
Leniolisib: a novel treatment for activated phosphoinositide-3 kinase delta syndrome.Front Pharmacol. 2024 Feb 12;15:1337436. doi: 10.3389/fphar.2024.1337436. eCollection 2024. Front Pharmacol. 2024. PMID: 38410131 Free PMC article.
-
Activated phosphoinositde 3-kinase (PI3Kδ) syndrome: an Italian point of view on diagnosis and new advances in treatment.Ital J Pediatr. 2024 May 20;50(1):103. doi: 10.1186/s13052-024-01662-5. Ital J Pediatr. 2024. PMID: 38769568 Free PMC article. Review.
-
Long-term treatment with selective PI3Kδ inhibitor leniolisib in adults with activated PI3Kδ syndrome.Blood Adv. 2024 Jun 25;8(12):3092-3108. doi: 10.1182/bloodadvances.2023011000. Blood Adv. 2024. PMID: 38593221 Free PMC article.
-
Modulating the PI3K Signalling Pathway in Activated PI3K Delta Syndrome: a Clinical Perspective.J Clin Immunol. 2023 Dec 27;44(1):34. doi: 10.1007/s10875-023-01626-0. J Clin Immunol. 2023. PMID: 38148368 Free PMC article. Review.
References
-
- Nunes-Santos CJ, Uzel G, Rosenzweig SD. PI3K pathway defects leading to immunodeficiency and immune dysregulation. J Allergy Clin Immunol. 2019;143(5):1676–87. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

