Peripheral neuropathy associated with monomethyl auristatin E-based antibody-drug conjugates
- PMID: 37727735
- PMCID: PMC10505985
- DOI: 10.1016/j.isci.2023.107778
Peripheral neuropathy associated with monomethyl auristatin E-based antibody-drug conjugates
Abstract
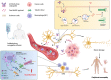
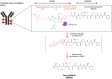
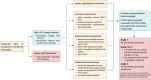
Since the successful approval of gemtuzumab ozogamicin, antibody-drug conjugates (ADCs) have emerged as a pivotal category of targeted therapies for cancer. Among these ADCs, the use of monomethyl auristatin E (MMAE) as a payload is prevalent in the development of ADC drugs, which has significantly improved overall therapeutic efficacy against various malignancies. However, increasing clinical observations have raised concerns regarding the potential nervous system toxicity associated with MMAE-based ADCs. Specifically, a higher incidence of peripheral neuropathy has been reported in ADCs incorporating MMAE as payloads. Considering the increasing global use of MMAE-based ADCs, it is imperative to provide an inclusive overview of diagnostic and management strategies for this adverse event. In this review, we examine current information and what future research directions are required to better understand and manage this type of clinical challenge.
Keywords: Cancer; Cancer systems biology; Health sciences; Medicine; Oncology; Toxicology.
© 2023 The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Microtubule and tubulin binding and regulation of microtubule dynamics by the antibody drug conjugate (ADC) payload, monomethyl auristatin E (MMAE): Mechanistic insights into MMAE ADC peripheral neuropathy.Toxicol Appl Pharmacol. 2021 Jun 15;421:115534. doi: 10.1016/j.taap.2021.115534. Epub 2021 Apr 20. Toxicol Appl Pharmacol. 2021. PMID: 33852878
-
Characterization of Site-Specifically Conjugated Monomethyl Auristatin E- and Duocarmycin-Based Anti-PSMA Antibody-Drug Conjugates for Treatment of PSMA-Expressing Tumors.J Nucl Med. 2018 Mar;59(3):494-501. doi: 10.2967/jnumed.117.196279. Epub 2017 Nov 16. J Nucl Med. 2018. PMID: 29146698
-
Development of a generalized pharmacokinetic model to characterize clinical pharmacokinetics of monomethyl auristatin E-based antibody-drug conjugates.Br J Clin Pharmacol. 2024 Jul;90(7):1637-1655. doi: 10.1111/bcp.16057. Epub 2024 Apr 2. Br J Clin Pharmacol. 2024. PMID: 38566392
-
Peripheral neuropathy with microtubule inhibitor containing antibody drug conjugates: Challenges and perspectives in translatability from nonclinical toxicology studies to the clinic.Regul Toxicol Pharmacol. 2016 Dec;82:1-13. doi: 10.1016/j.yrtph.2016.10.012. Epub 2016 Oct 20. Regul Toxicol Pharmacol. 2016. PMID: 27773754 Review.
-
Clinical toxicity of antibody drug conjugates: a meta-analysis of payloads.Invest New Drugs. 2018 Feb;36(1):121-135. doi: 10.1007/s10637-017-0520-6. Epub 2017 Oct 13. Invest New Drugs. 2018. PMID: 29027591 Review.
Cited by
-
The Vedotin Antibody-Drug Conjugate Payload Drives Platform-Based Nonclinical Safety and Pharmacokinetic Profiles.Mol Cancer Ther. 2024 Oct 1;23(10):1483-1493. doi: 10.1158/1535-7163.MCT-24-0087. Mol Cancer Ther. 2024. PMID: 38692647 Free PMC article.
-
A Head-to-Head Comparison of the First-Line Treatments for Locally Advanced or Metastatic Urothelial Cancer: Is There Still a Role for Chemotherapy?Cancers (Basel). 2024 Jun 29;16(13):2400. doi: 10.3390/cancers16132400. Cancers (Basel). 2024. PMID: 39001462 Free PMC article.
-
Targeted Delivery Strategies for Multiple Myeloma and Their Adverse Drug Reactions.Pharmaceuticals (Basel). 2024 Jun 25;17(7):832. doi: 10.3390/ph17070832. Pharmaceuticals (Basel). 2024. PMID: 39065683 Free PMC article. Review.
-
Bystander Effects, Pharmacokinetics, and Linker-Payload Stability of EGFR-Targeting Antibody-Drug Conjugates Losatuxizumab Vedotin and Depatux-M in Glioblastoma Models.Clin Cancer Res. 2024 Aug 1;30(15):3287-3297. doi: 10.1158/1078-0432.CCR-24-0426. Clin Cancer Res. 2024. PMID: 38743766 Free PMC article.
References
-
- Akaiwa M., Dugal-Tessier J., Mendelsohn B.A. Antibody-drug conjugate payloads; study of auristatin derivatives. Chem. Pharm. Bull. 2020;68:201–211. - PubMed
-
- Bartlett N.L., Sharman J.P., Oki Y., Advani R.H., Bello C.M., Winter J.N., Yang Y., Kennedy D.A., Jacobsen E.D. A phase 2 study of brentuximab vedotin in patients with relapsed or refractory CD30-positive non-Hodgkin lymphomas: interim results in patients with DLBCL and other B-cell lymphomas. Blood. 2013;122:848.
-
- Pro B., Advani R.H., Brice P., Bartlett N.L., Rosenblatt J.D., Illidge T., Matous J., Ramchandren R., Fanale M.A., Connors J.M., et al. Three-year survival results from an ongoing phase 2 study of brentuximab vedotin in patients with relapsed or refractory systemic anaplastic large cell lymphoma. Blood. 2013;122:1809. - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources

