Long-term efficacy and safety of osilodrostat in patients with Cushing's disease: results from the LINC 4 study extension
- PMID: 37680892
- PMCID: PMC10482037
- DOI: 10.3389/fendo.2023.1236465
Long-term efficacy and safety of osilodrostat in patients with Cushing's disease: results from the LINC 4 study extension
Abstract
Objective: To evaluate the long-term efficacy and safety of osilodrostat in patients with Cushing's disease.
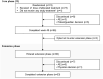
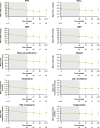
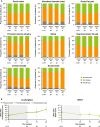
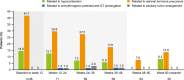
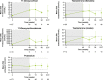
Methods: The multicenter, 48-week, Phase III LINC 4 clinical trial had an optional extension period that was initially intended to continue to week 96. Patients could continue in the extension until a managed-access program or alternative treatment became available locally, or until a protocol amendment was approved at their site that specified that patients should come for an end-of-treatment visit within 4 weeks or by week 96, whichever occurred first. Study outcomes assessed in the extension included: mean urinary free cortisol (mUFC) response rates; changes in mUFC, serum cortisol and late-night salivary cortisol (LNSC); changes in cardiovascular and metabolic-related parameters; blood pressure, waist circumference and weight; changes in physical manifestations of Cushing's disease; changes in patient-reported outcomes for health-related quality of life; changes in tumor volume; and adverse events. Results were analyzed descriptively; no formal statistical testing was performed.
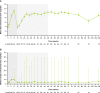
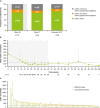
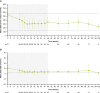
Results: Of 60 patients who entered, 53 completed the extension, with 29 patients receiving osilodrostat for more than 96 weeks (median osilodrostat duration: 87.1 weeks). The proportion of patients with normalized mUFC observed in the core period was maintained throughout the extension. At their end-of-trial visit, 72.4% of patients had achieved normal mUFC. Substantial reductions in serum cortisol and LNSC were also observed. Improvements in most cardiovascular and metabolic-related parameters, as well as physical manifestations of Cushing's disease, observed in the core period were maintained or continued to improve in the extension. Osilodrostat was generally well tolerated; the safety profile was consistent with previous reports.
Conclusion: Osilodrostat provided long-term control of cortisol secretion that was associated with sustained improvements in clinical signs and physical manifestations of hypercortisolism. Osilodrostat is an effective long-term treatment for patients with Cushing's disease.
Clinical trial registration: ClinicalTrials.gov, identifier NCT02180217.
Keywords: 11β-hydroxylase; Cushing’s disease; hypercortisolism; long-term treatment; osilodrostat.
Copyright © 2023 Gadelha, Snyder, Witek, Bex, Belaya, Turcu, Feelders, Heaney, Paul, Pedroncelli and Auchus.
Conflict of interest statement
Author MG has received speaker fees from Recordati, Ipsen, Crinetics Pharmaceuticals, and Novo Nordisk and attended advisory boards for Novo Nordisk, Recordati, Ipsen, and Crinetics Pharmaceuticals. Author PS reports consultancy for Teva Pharmaceuticals. Author PW reports receiving travel grants and speaker fees from Novartis, Ipsen, Recordati, Novo Nordisk, Strongbridge Biopharma now Xeris Pharmaceuticals, and Lilly. Author MB reports receiving travel grants from Novartis, Ipsen, and Pfizer and consultancy for Novartis. Author ZB has nothing to disclose. Author AT reports consultancy for CinCor and PhaseBio. Author RF reports consultancy for HRA Pharma and Recordati and a research grant from Corcept Therapeutics. Author AH reports speaker fees from Chiasma and Ipsen and has been an advisor to Strongbridge Biopharma now Xeris Pharmaceuticals, Novo Nordisk, and Lundbeck Pharma. Author MP is employed by the company Novartis Pharma AG. Author AP was employed by the company Recordati AG at the time of manuscript development. Author RA reports grants and personal fees from Xeris Pharmaceuticals, Spruce Biosciences, Neurocrine Biosciences, Corcept Therapeutics, Diurnal Ltd, Sparrow Pharmaceuticals, and Novartis and personal fees from Adrenas Therapeutics, Janssen Pharmaceuticals, Quest Diagnostics, Crinetics Pharmaceuticals, PhaseBio Pharmaceuticals, H Lundbeck A/S, Novo Nordisk, and Recordati Rare Diseases.
Figures
Similar articles
-
Efficacy and safety of osilodrostat in patients with Cushing's disease (LINC 3): a multicentre phase III study with a double-blind, randomised withdrawal phase.Lancet Diabetes Endocrinol. 2020 Sep;8(9):748-761. doi: 10.1016/S2213-8587(20)30240-0. Epub 2020 Jul 27. Lancet Diabetes Endocrinol. 2020. PMID: 32730798 Clinical Trial.
-
Long-term efficacy and safety of osilodrostat in Cushing's disease: final results from a Phase II study with an optional extension phase (LINC 2).Pituitary. 2022 Dec;25(6):959-970. doi: 10.1007/s11102-022-01280-6. Epub 2022 Oct 11. Pituitary. 2022. PMID: 36219274 Free PMC article. Clinical Trial.
-
Improvement in clinical features of hypercortisolism during osilodrostat treatment: findings from the Phase III LINC 3 trial in Cushing's disease.J Endocrinol Invest. 2024 Oct;47(10):2437-2448. doi: 10.1007/s40618-024-02359-6. Epub 2024 May 2. J Endocrinol Invest. 2024. PMID: 38696122 Free PMC article. Clinical Trial.
-
Clinical Utility of Osilodrostat in Cushing's Disease: Review of Currently Available Literature.Drug Des Devel Ther. 2023 Apr 27;17:1303-1312. doi: 10.2147/DDDT.S315359. eCollection 2023. Drug Des Devel Ther. 2023. PMID: 37143705 Free PMC article. Review.
-
Osilodrostat for the treatment of Cushing's disease.Expert Opin Pharmacother. 2021 Jun;22(9):1099-1106. doi: 10.1080/14656566.2021.1897106. Epub 2021 Mar 11. Expert Opin Pharmacother. 2021. PMID: 33703978 Review.
Cited by
-
Current and Emerging Pharmacological Therapies for Cushing's Disease.Curr Pharm Des. 2024;30(10):757-777. doi: 10.2174/0113816128290025240216110928. Curr Pharm Des. 2024. PMID: 38424426 Review.
-
Efficacy and Safety of Osilodrostat in Managing Cushing's Syndrome: A Systematic Review and Meta-Analysis.Indian J Endocrinol Metab. 2024 May-Jun;28(3):232-238. doi: 10.4103/ijem.ijem_260_23. Epub 2024 Jun 26. Indian J Endocrinol Metab. 2024. PMID: 39086571 Free PMC article. Review.
-
Cushing's Disease: Long-Term Effectiveness and Safety of Osilodrostat in a Polish Group of Patients with Persistent Hypercortisolemia in the Experience of a Single Center.Biomedicines. 2023 Dec 6;11(12):3227. doi: 10.3390/biomedicines11123227. Biomedicines. 2023. PMID: 38137448 Free PMC article.

