Belantamab Mafodotin: From Clinical Trials Data to Real-Life Experiences
- PMID: 37296910
- PMCID: PMC10251850
- DOI: 10.3390/cancers15112948
Belantamab Mafodotin: From Clinical Trials Data to Real-Life Experiences
Abstract
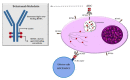
Despite the recent approval of novel immunotherapies, such as immunomodulatory drugs, proteasome inhibitors and anti-CD38 monoclonal antibodies, Multiple Myeloma (MM) remains incurable, and the acquisition of triple-refractoriness leads to really dismal outcomes in even earlier lines of therapy. More recently, innovative therapeutic strategies targeting B cell maturation antigen (BCMA), highly expressed on the plasma cell surface, are drawing different future landscapes in terms of effectiveness and outcomes. Belantamab Mafodotin, a first-in-class anti-BCMA antibody-drug conjugate, demonstrated good efficacy and safety profile in triple-refractory patients in the phase 2 DREAMM-2 trial, and it was approved for the treatment of MM triple-exposed patients with >4 prior lines of therapy. Here, starting from Belantamab Mafodotin clinical trials and also exploring combination studies and different schedules in order to improve its efficacy and toxicity, we focused on real-life experiences all over the world, which have confirmed clinical trial data and encourage further Belantamab Mafodotin investigations.
Keywords: antibody–drug conjugate; belantamab mafodotin; multiple myeloma.
Conflict of interest statement
S.M.: Honoraria from Amgen, Janssen. L.C.: Honoraria from Celgene, Janssen, Amgen; M.O.: Honoraria from and Advisor for AbbVie, Amgen, BMS, Celgene, GSK, Janssen, Roche, Sanofi, Takeda; M.T.P.: Honoraria from Janssen-Cilag, Celgene-BMS, Amgen, Sanofi, GSK, Takeda; Advisor for Janssen-Cilag, Celgene-BMS, Amgen, Sanofi, GSK, Takeda, Roche, Karyopharm; Support for attending meetings and/or travel from Janssen-Cilag, Celgene-BMS, Amgen, Sanofi, Takeda. F.F.: Advisor for Takeda, Sanofi, Honoraria from GSK, Janssen, Amgen.
Figures
Similar articles
-
Belantamab mafodotin for the treatment of multiple myeloma.Drugs Today (Barc). 2021 Nov;57(11):653-663. doi: 10.1358/dot.2021.57.11.3319146. Drugs Today (Barc). 2021. PMID: 34821879
-
Belantamab Mafodotin for the Treatment of Multiple Myeloma: An Overview of the Clinical Efficacy and Safety.Drug Des Devel Ther. 2021 Jun 2;15:2401-2415. doi: 10.2147/DDDT.S267404. eCollection 2021. Drug Des Devel Ther. 2021. PMID: 34103900 Free PMC article. Review.
-
EMA Review of Belantamab Mafodotin (Blenrep) for the Treatment of Adult Patients with Relapsed/Refractory Multiple Myeloma.Oncologist. 2021 Jan;26(1):70-76. doi: 10.1002/onco.13592. Epub 2020 Nov 23. Oncologist. 2021. PMID: 33179377 Free PMC article. Clinical Trial.
-
Exposure-Response Analyses for Therapeutic Dose Selection of Belantamab Mafodotin in Patients With Relapsed/Refractory Multiple Myeloma.Clin Pharmacol Ther. 2021 Nov;110(5):1282-1292. doi: 10.1002/cpt.2409. Epub 2021 Oct 4. Clin Pharmacol Ther. 2021. PMID: 34468979 Free PMC article. Clinical Trial.
-
The role of belantamab mafodotin for patients with relapsed and/or refractory multiple myeloma.Ther Adv Hematol. 2020 Dec 14;11:2040620720979813. doi: 10.1177/2040620720979813. eCollection 2020. Ther Adv Hematol. 2020. PMID: 33403093 Free PMC article. Review.
Cited by
-
Cardiovascular adverse events associated with targeted therapies for multiple myeloma: a pharmacovigilance study.Front Immunol. 2024 Sep 26;15:1400101. doi: 10.3389/fimmu.2024.1400101. eCollection 2024. Front Immunol. 2024. PMID: 39391316 Free PMC article.
-
Real-World Effectiveness and Safety of Belantamab Mafodotin Monotherapy in Triple-Class Refractory Multiple Myeloma.Int J Mol Sci. 2023 Jul 23;24(14):11829. doi: 10.3390/ijms241411829. Int J Mol Sci. 2023. PMID: 37511588 Free PMC article.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

