Curcumin-Functionalized Graphene Oxide Strongly Prevents Candida parapsilosis Adhesion and Biofilm Formation
- PMID: 37259419
- PMCID: PMC9967767
- DOI: 10.3390/ph16020275
Curcumin-Functionalized Graphene Oxide Strongly Prevents Candida parapsilosis Adhesion and Biofilm Formation
Abstract
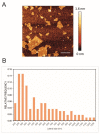
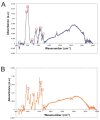
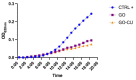
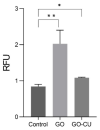
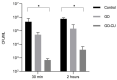
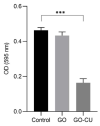
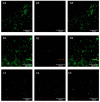
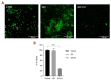
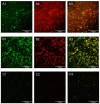
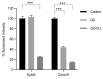
Candida parapsilosis is the major non-C. albicans species involved in the colonization of central venous catheters, causing bloodstream infections. Biofilm formation on medical devices is considered one of the main causes of healthcare-associated infections and represents a global public health problem. In this context, the development of new nanomaterials that exhibit anti-adhesive and anti-biofilm properties for the coating of medical devices is crucial. In this work, we aimed to characterize the antimicrobial activity of two different coated-surfaces, graphene oxide (GO) and curcumin-graphene oxide (GO/CU) for the first time, against C. parapsilosis. We report the capacity of GO to bind and stabilize CU molecules, realizing a homogenous coated surface. We tested the anti-planktonic activity of GO and GO/CU by growth curve analysis and quantification of Reactive Oxigen Species( ROS) production. Then, we tested the antibiofilm activity by adhesion assay, crystal violet assay, and live and dead assay; moreover, the inhibition of the formation of a mature biofilm was investigated by a viability test and the use of specific dyes for the visualization of the cells and the extra-polymeric substances. Our data report that GO/CU has anti-planktonic, anti-adhesive, and anti-biofilm properties, showing a 72% cell viability reduction and a decrease of 85% in the secretion of extra-cellular substances (EPS) after 72 h of incubation. In conclusion, we show that the GO/CU conjugate is a promising material for the development of medical devices that are refractory to microbial colonization, thus leading to a decrease in the impact of biofilm-related infections.
Keywords: biofilm; biofilm-related infections; graphene; natural anti-biofilm compounds.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
In vitro antifungal activity and in vivo antibiofilm activity of cerium nitrate against Candida species.J Antimicrob Chemother. 2015 Apr;70(4):1083-93. doi: 10.1093/jac/dku511. Epub 2015 Jan 3. J Antimicrob Chemother. 2015. PMID: 25558078
-
Graphene oxide coatings prevent Candida albicans biofilm formation with a controlled release of curcumin-loaded nanocomposites.Nanomedicine (Lond). 2018 Nov;13(22):2867-2879. doi: 10.2217/nnm-2018-0183. Epub 2018 Nov 15. Nanomedicine (Lond). 2018. PMID: 30431405
-
Anti-Candida activity assessment of Pelargonium graveolens oil free and nanoemulsion in biofilm formation in hospital medical supplies.Microb Pathog. 2016 Nov;100:170-178. doi: 10.1016/j.micpath.2016.08.013. Epub 2016 Aug 18. Microb Pathog. 2016. PMID: 27544324
-
Graphene Oxide Coatings as Tools to Prevent Microbial Biofilm Formation on Medical Device.Adv Exp Med Biol. 2020;1282:21-35. doi: 10.1007/5584_2019_434. Adv Exp Med Biol. 2020. PMID: 31468360 Review.
-
Graphene-Based Materials for Inhibition of Wound Infection and Accelerating Wound Healing.Biomed Pharmacother. 2023 Feb;158:114184. doi: 10.1016/j.biopha.2022.114184. Epub 2022 Dec 30. Biomed Pharmacother. 2023. PMID: 36587554 Review.
Cited by
-
Graphene Oxide Nanostructures as Nanoplatforms for Delivering Natural Therapeutic Agents: Applications in Cancer Treatment, Bacterial Infections, and Bone Regeneration Medicine.Nanomaterials (Basel). 2023 Sep 28;13(19):2666. doi: 10.3390/nano13192666. Nanomaterials (Basel). 2023. PMID: 37836307 Free PMC article. Review.
-
Comparison of Antimicrobial Properties of Graphene Oxide-Based Materials, Carbon Dots, and Their Combinations Deposited on Cotton Fabrics.Int J Mol Sci. 2024 May 14;25(10):5328. doi: 10.3390/ijms25105328. Int J Mol Sci. 2024. PMID: 38791366 Free PMC article.
-
Photodynamic impact of curcumin enhanced silver functionalized graphene nanocomposites on Candida virulence.Discov Nano. 2024 Apr 29;19(1):71. doi: 10.1186/s11671-024-04017-5. Discov Nano. 2024. PMID: 38683264 Free PMC article.
-
Died or Not Dyed: Assessment of Viability and Vitality Dyes on Planktonic Cells and Biofilms from Candida parapsilosis.J Fungi (Basel). 2024 Mar 11;10(3):209. doi: 10.3390/jof10030209. J Fungi (Basel). 2024. PMID: 38535217 Free PMC article.
-
An Overview of Biofilm-Associated Infections and the Role of Phytochemicals and Nanomaterials in Their Control and Prevention.Pharmaceutics. 2024 Jan 24;16(2):162. doi: 10.3390/pharmaceutics16020162. Pharmaceutics. 2024. PMID: 38399223 Free PMC article. Review.
References
-
- Bugli F., Posteraro B., Papi M., Torelli R., Maiorana A., Sterbini F.P., Posteraro P., Sanguinetti M., de Spirito M. In Vitro Interaction between Alginate Lyase and Amphotericin B against Aspergillus Fumigatus Biofilm Determined by Different Methods. Antimicrob. Agents Chemother. 2013;57:1275–1282. doi: 10.1128/AAC.01875-12. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

