Tisotumab Vedotin Safety and Tolerability in Clinical Practice: Managing Adverse Events
- PMID: 37009403
- PMCID: PMC10062530
- DOI: 10.6004/jadpro.2023.14.2.4
Tisotumab Vedotin Safety and Tolerability in Clinical Practice: Managing Adverse Events
Abstract
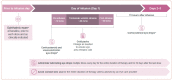
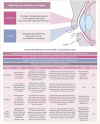
Tisotumab vedotin-tftv, an antibody-drug conjugate indicated for the treatment of adult patients with recurrent or metastatic cervical cancer with disease progression on or after chemotherapy, demonstrated clinically meaningful and durable responses with a manageable safety profile in the pivotal phase II innovaTV 204 clinical trial. Based on the proposed mechanism of action of tisotumab vedotin, experience from clinical trials, and the US prescribing information, certain adverse events (AEs) including ocular AEs, peripheral neuropathy, and bleeding have been identified as AEs of interest. This article highlights practical considerations and provides recommendations to support the management of selected AEs associated with tisotumab vedotin. Central to monitoring of patients on tisotumab vedotin is a comprehensive care team comprised of oncologists, advanced practice providers (including nurse practitioners, physician assistants, and pharmacists), and other specialists such as ophthalmologists. As ocular AEs may be less familiar to gynecologic oncology practitioners, adherence to the "Premedication and Required Eye Care" section outlined in the US prescribing information, as well as the incorporation of ophthalmologists into the oncology care team, can help provide timely and appropriate eye care for patients receiving tisotumab vedotin.
© 2023 Harborside™.
Conflict of interest statement
The authors have no conflicts of interest to disclose.
Figures
Similar articles
-
Mitigation and management strategies for ocular events associated with tisotumab vedotin.Gynecol Oncol. 2022 May;165(2):385-392. doi: 10.1016/j.ygyno.2022.02.010. Epub 2022 Mar 8. Gynecol Oncol. 2022. PMID: 35277279 Review.
-
Tisotumab vedotin in patients with advanced or metastatic solid tumours (InnovaTV 201): a first-in-human, multicentre, phase 1-2 trial.Lancet Oncol. 2019 Mar;20(3):383-393. doi: 10.1016/S1470-2045(18)30859-3. Epub 2019 Feb 8. Lancet Oncol. 2019. PMID: 30745090 Clinical Trial.
-
A review of the novel tissue factor antibody-drug conjugate: Tisotumab vedotin.J Oncol Pharm Pract. 2023 Mar;29(2):441-449. doi: 10.1177/10781552221139775. Epub 2022 Nov 22. J Oncol Pharm Pract. 2023. PMID: 36415085 Review.
-
Efficacy and safety of tisotumab vedotin in previously treated recurrent or metastatic cervical cancer (innovaTV 204/GOG-3023/ENGOT-cx6): a multicentre, open-label, single-arm, phase 2 study.Lancet Oncol. 2021 May;22(5):609-619. doi: 10.1016/S1470-2045(21)00056-5. Epub 2021 Apr 9. Lancet Oncol. 2021. PMID: 33845034 Clinical Trial.
-
A Review of Tisotumab Vedotin-tftv in Recurrent or Metastatic Cervical Cancer.Ann Pharmacother. 2023 May;57(5):585-596. doi: 10.1177/10600280221118370. Epub 2022 Aug 12. Ann Pharmacother. 2023. PMID: 35962528 Review.
Cited by
-
Introduction of Carbonyl Groups into Antibodies.Molecules. 2023 Dec 1;28(23):7890. doi: 10.3390/molecules28237890. Molecules. 2023. PMID: 38067618 Free PMC article. Review.
-
Incidence and Mitigation of Corneal Pseudomicrocysts Induced by Antibody-Drug Conjugates (ADCs).Curr Ophthalmol Rep. 2024 Jun;12(2):13-22. doi: 10.1007/s40135-024-00322-5. Epub 2024 Mar 24. Curr Ophthalmol Rep. 2024. PMID: 38756824 Free PMC article.
References
-
- Akpek, E. K., & Smith, R. A. (2013). Overview of age-related ocular conditions. American Journal of Managed Care, 19(5 Suppl), S67–S75. https://www.ajmc.com/view/ace011_13may_agingeye_akpek - PubMed
-
- Alley, S. C., Harris, J. R., Cao, A., Heuvel, E. G.-v. d., Velayudhan, J., Satijn,…Breij, E. C. (2019). Abstract 221: Tisotumab vedotin induces anti-tumor activity through MMAE-mediated, Fc-mediated, and Fab-mediated effector functions in vitro. Cancer Research, 79(13 Supplement), 221–221. 10.1158/1538-7445.AM2019-221 - DOI
-
- Amescua, G., Akpek, E. K., Farid, M., Garcia-Ferrer, F. J., Lin, A., Rhee, M. K.,…American Academy of Ophthalmology Preferred Practice Pattern Cornea and External Disease Panel. (2019). Blepharitis preferred practice pattern®. Ophthalmology, 126(1), P56–P93. 10.1016/j.ophtha.2018.10.019 - DOI - PubMed
-
- Ancuţa, B., Nemes, R., Scurtu, S., Shencker, M., & Iliescu, D. (2012). Quality of life in cervical cancer survivors. Acta Medica Marisiensis, 58(5), 275–277.
-
- Ando, R., Kase, S., Ohashi, T., Dong, Z., Fukuhara, J., Kanda, A.,…Ishida, S. (2011). Tissue factor expression in human pterygium. Molecular Vision, 17, 63–69. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3021580/ - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
