PPAR agonists for the treatment of primary biliary cholangitis: Old and new tales
- PMID: 36684809
- PMCID: PMC9850184
- DOI: 10.1016/j.jtauto.2023.100188
PPAR agonists for the treatment of primary biliary cholangitis: Old and new tales
Abstract
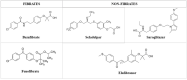
Introduction: Primary biliary cholangitis (PBC) is an autoimmune liver disease involving the small intrahepatic bile ducts; when untreated or undertreated, it may evolve to liver fibrosis and cirrhosis. Ursodeoxycholic Acid (UDCA) is the standard of care treatment, Obeticholic Acid (OCA) has been approved as second-line therapy for those non responder or intolerant to UDCA. However, due to moderate rate of UDCA-non responders and to warnings recently issued against OCA use in patients with cirrhosis, further therapies are needed.Areas covered. Deep investigations into the pathogenesis of PBC is leading to proposal of new therapeutic agents, among which peroxisome proliferator-activated receptor (PPAR) ligands seem to be highly promising given the preliminary, positive results in Phase 2 and 3 trials. Bezafibrate, the most evaluated, is currently used in clinical practice in combination with UDCA in referral centers. We herein describe completed and ongoing trials involving PPAR agonists use in PBC, analyzing pits and falls.
Expert opinion: Testing new therapeutic opportunities in PBC is challenging due to its low prevalence and slow progression. However, new drugs including PPAR agonists, are currently under investigation and should be considered for at-risk PBC patients.
Keywords: AEs, adverse events; AIH, Autoimmune Hepatitis; ALP, Alkaline Phosphatase; AMA, Antimitochondrial antibodies; BZF, Bezafibrate; CKD, chronic kidney disease; Elafibranor; FDA, Food and Drug; FF, Fenofibrate; FXR, Farnesoid X Receptor; Fibrates; GGT, γ-glutamil transferase; HCC, Hepatocellular Carcinoma; HDL, high-density lipoprotein; HR, Hazard Ratio; HSC, Hepatic Stellate Cells; IL-1β, Interleukin-1; IgM, Immunoglobulin M; LDL, low-density- lipoprotein; LT, Liver Transplant; MDR3, multidrug resistance protein 3; NASH, Non Alcoholic Steato-Hepatits; NRS, Numerical Raing Scale; OCA, Obeticholic Acid; OR, Odds Ratio; PAR, protease-activated receptors; PBC, Primary Biliary Cholangitis; PC, phosphatidylcholine; PH, Portal Hypertension; PPAR agonists; PPAR, peroxisome proliferator-activated receptor; Primary biliary cholangitis; QoL, Quality of Life; RCT, randomized controlled trial; SAE, Severe Adverse Event; Saroglitazar; Seladelpar; TGR, transmembrane G protein-coupled receptor; TLR, Toll Like Receptor; TNF-α, Tumor Necrosis Factor- α; UDCA; UDCA, ursodeoxycholic acid; UK, United Kingdom; ULN, upper limit of normal; VAS, Visual Analogue Scale; VRS, Verbal Rating Scale.
© 2023 The Authors.
Conflict of interest statement
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests:Ana Lleo reports a relationship with Gilead Sciences Inc that includes: speaking and lecture fees. Ana Lleo reports a relationship with Intercept Pharmaceuticals that includes: consulting or advisory and speaking and lecture fees. Ana Lleo reports a relationship with Alfasigma SpA that includes: consulting or advisory and speaking and lecture fees. Ana Lleo reports a relationship with Albireo Pharma Inc that includes: consulting or advisory. Ana Lleo reports a relationship with GSK that includes: speaking and lecture fees. Ana Lleo reports a relationship with AstraZeneca Pharmaceuticals LP that includes: consulting or advisory. Ana Lleo reports a relationship with Incyte Corporation that includes: speaking and lecture fees. Ana Lleo reports a relationship with Takeda Pharmaceutical Co Ltd that includes: consulting or advisory. Ana Lleo reports a relationship with Kowa Pharmaceutical Europe Co Ltd that includes: consulting or advisory. Ana Lleo reports a relationship with Merck Sharp & Dohme UK Ltd that includes: speaking and lecture fees. Ana Lleo reports a relationship with AbbVie Inc that includes: speaking and lecture fees. Francesca Colapietro reports a relationship with Intercept Pharmaceuticals Inc that includes: speaking and lecture fees. Research Grants: EU Project D-LIVER, EU COST Action EURO-Cholangio-Net, Italian Ministry of Health, Italian Association for Cancer Research (AIRC) Support for sponsored studies (via Humanitas Research Hospital): Falk, Intercept Pharma.
Figures
Similar articles
-
Obeticholic acid for the treatment of primary biliary cholangitis.Expert Opin Pharmacother. 2016 Sep;17(13):1809-15. doi: 10.1080/14656566.2016.1218471. Epub 2016 Aug 9. Expert Opin Pharmacother. 2016. PMID: 27468093 Review.
-
Farnesoid X receptor agonist tropifexor attenuates cholestasis in a randomised trial in patients with primary biliary cholangitis.JHEP Rep. 2022 Jul 21;4(11):100544. doi: 10.1016/j.jhepr.2022.100544. eCollection 2022 Nov. JHEP Rep. 2022. PMID: 36267872 Free PMC article.
-
Real-world experience with obeticholic acid in patients with primary biliary cholangitis.JHEP Rep. 2021 Jan 27;3(2):100248. doi: 10.1016/j.jhepr.2021.100248. eCollection 2021 Apr. JHEP Rep. 2021. PMID: 33681748 Free PMC article.
-
Update on New Drugs and Those in Development for the Treatment of Primary Biliary Cholangitis.Gastroenterol Hepatol (N Y). 2018 Mar;14(3):154-163. Gastroenterol Hepatol (N Y). 2018. PMID: 29928160 Free PMC article.
-
Obeticholic acid-a new therapy in PBC and NASH.Br Med Bull. 2020 May 15;133(1):95-104. doi: 10.1093/bmb/ldaa006. Br Med Bull. 2020. PMID: 32282030 Review.
Cited by
-
PPARs in Clinical Experimental Medicine after 35 Years of Worldwide Scientific Investigations and Medical Experiments.Biomolecules. 2024 Jul 1;14(7):786. doi: 10.3390/biom14070786. Biomolecules. 2024. PMID: 39062500 Free PMC article. Review.
-
The treatment of primary biliary cholangitis: from shadow to light.Therap Adv Gastroenterol. 2024 Jul 29;17:17562848241265782. doi: 10.1177/17562848241265782. eCollection 2024. Therap Adv Gastroenterol. 2024. PMID: 39081664 Free PMC article. Review.
-
Efficacy and safety of peroxisome proliferator-activated receptor agonists for the treatment of primary biliary cholangitis: a meta-analysis of randomized controlled trials.Front Pharmacol. 2024 Jul 22;15:1432814. doi: 10.3389/fphar.2024.1432814. eCollection 2024. Front Pharmacol. 2024. PMID: 39108746 Free PMC article. Review.
-
Current Therapies for Cholestatic Diseases.Biomedicines. 2023 Jun 15;11(6):1713. doi: 10.3390/biomedicines11061713. Biomedicines. 2023. PMID: 37371808 Free PMC article. Review.
-
Elafibranor: A promising treatment for alcohol-associated liver disease?World J Gastroenterol. 2024 Oct 21;30(39):4313-4317. doi: 10.3748/wjg.v30.i39.4313. World J Gastroenterol. 2024. PMID: 39492824 Free PMC article.
References
-
- European e. e. e. Association for the study of the liver. Electronic address, L. European association for the study of the. EASL clinical practice guidelines: the diagnosis and management of patients with primary biliary cholangitis. J. Hepatol. 2017;67:145–172. doi: 10.1016/j.jhep.2017.03.022. - DOI - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous