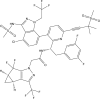
Lenacapavir: First Approval
- PMID: 36272024
- PMCID: PMC10267266
- DOI: 10.1007/s40265-022-01786-0
Lenacapavir: First Approval
Erratum in
-
Correction to: Lenacapavir: First Approval.Drugs. 2023 Jul;83(11):1061. doi: 10.1007/s40265-023-01908-2. Drugs. 2023. PMID: 37314634 Free PMC article. No abstract available.
Abstract
Lenacapavir (Sunlenca®) is a long-acting capsid inhibitor of human immunodeficiency virus type 1 (HIV-1) being developed by Gilead Sciences Inc. It is available as an oral tablet and injectable solution, with the latter being a slow-release formulation to allow bi-annual subcutaneous administration. In August 2022, lenacapavir received its first approval in the EU for use in combination with other antiretroviral(s) in adults with multi-drug resistant HIV infection, for whom it is otherwise not possible to construct a suppressive anti-viral regimen. This article summarizes the milestones in the development of lenacapavir leading to this first approval for the treatment of HIV-1 infection.
© 2022. The Author(s), under exclusive licence to Springer Nature Switzerland AG.
Conflict of interest statement
During the peer review process the manufacturer of the agent under review was offered an opportunity to comment on the article. Changes resulting from any comments received were made by the authors on the basis of scientific completeness and accuracy. Julia Paik is a salaried employee of Adis International Ltd/Springer Nature, and declares no relevant conflicts of interest. All authors contributed to the review and are responsible for the article content.
Figures
Similar articles
-
Lenacapavir: A first-in-class capsid inhibitor for the treatment of highly treatment-resistant HIV.Am J Health Syst Pharm. 2023 Dec 5;80(24):1774-1780. doi: 10.1093/ajhp/zxad223. Am J Health Syst Pharm. 2023. PMID: 37767713 Review.
-
Lenacapavir: A novel injectable HIV-1 capsid inhibitor.Int J Antimicrob Agents. 2024 Jan;63(1):107009. doi: 10.1016/j.ijantimicag.2023.107009. Epub 2023 Oct 14. Int J Antimicrob Agents. 2024. PMID: 37844807 Review.
-
Efficacy and safety of the novel capsid inhibitor lenacapavir to treat multidrug-resistant HIV: week 52 results of a phase 2/3 trial.Lancet HIV. 2023 Aug;10(8):e497-e505. doi: 10.1016/S2352-3018(23)00113-3. Epub 2023 Jul 11. Lancet HIV. 2023. PMID: 37451297 Clinical Trial.
-
Highlights on the Development, Related Patents, and Prospects of Lenacapavir: The First-in-Class HIV-1 Capsid Inhibitor for the Treatment of Multi-Drug-Resistant HIV-1 Infection.Medicina (Kaunas). 2023 May 28;59(6):1041. doi: 10.3390/medicina59061041. Medicina (Kaunas). 2023. PMID: 37374245 Free PMC article. Review.
-
Lenacapavir: A Novel Long-Acting Capsid Inhibitor for HIV.Ann Pharmacother. 2024 Feb;58(2):185-195. doi: 10.1177/10600280231171375. Epub 2023 May 3. Ann Pharmacother. 2024. PMID: 37138515 Review.
Cited by
-
Pyrazole: an emerging privileged scaffold in drug discovery.Future Med Chem. 2023 Nov;15(21):2011-2023. doi: 10.4155/fmc-2023-0207. Epub 2023 Nov 7. Future Med Chem. 2023. PMID: 37933613 Free PMC article. Review.
-
Educational Review: Neoadjuvant Approaches to Melanoma.Ann Surg Oncol. 2022 Dec;29(13):8492-8500. doi: 10.1245/s10434-022-12224-6. Epub 2022 Jul 18. Ann Surg Oncol. 2022. PMID: 35849287 Review.
-
Chronic Hepatitis B Infection: New Approaches towards Cure.Biomolecules. 2023 Aug 1;13(8):1208. doi: 10.3390/biom13081208. Biomolecules. 2023. PMID: 37627273 Free PMC article. Review.
-
Antiretroviral drugs from multiple classes induce loss of excitatory synapses between hippocampal neurons in culture.Front Pharmacol. 2024 Mar 12;15:1369757. doi: 10.3389/fphar.2024.1369757. eCollection 2024. Front Pharmacol. 2024. PMID: 38533258 Free PMC article.
-
Lack of Resistance Mutations to the Novel HIV-1 Capsid Inhibitor Lenacapavir Among People Living with HIV in Guangdong, China.Infect Drug Resist. 2024 Oct 2;17:4271-4277. doi: 10.2147/IDR.S484383. eCollection 2024. Infect Drug Resist. 2024. PMID: 39377030 Free PMC article.
References
-
- Gilead Sciences Inc. Lenacapavir (Sunlenca®): EU summary of product characteristics. 2022. https://ema.europa.eu/. Accessed 21 Sep 2022.
-
- Gilead Sciences Inc. Gilead announces first regulatory approval for Sunlenca® (lenacapavir), the only twice-yearly HIV treatment option [media release]. 22 Aug 2022. https://www.gilead.com/.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical