Natural history of Type 1 spinal muscular atrophy: a retrospective, global, multicenter study
- PMID: 35906608
- PMCID: PMC9336055
- DOI: 10.1186/s13023-022-02455-x
Natural history of Type 1 spinal muscular atrophy: a retrospective, global, multicenter study
Abstract
Background: ANCHOVY was a global, multicenter, chart-review study that aimed to describe the natural history of Type 1 spinal muscular atrophy (SMA) from a broad geographical area and provide further contextualization of results from the FIREFISH (NCT02913482) interventional study of risdiplam treatment in Type 1 SMA.
Methods: Data were extracted from medical records of patients with first symptoms attributable to Type 1 SMA between 28 days and 3 months of age, genetic confirmation of SMA, and confirmed survival of motor neuron 2 copy number of two or unknown. The study period started on 1 January 2008 for all sites; study end dates were site-specific due to local treatment availabilities. Primary endpoints were time to death and/or permanent ventilation and proportion of patients achieving motor milestones. Secondary endpoints included time to initiation of respiratory and feeding support.
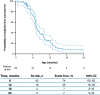
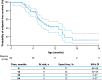
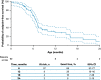
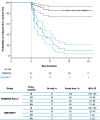
Results: Data for 60 patients from nine countries across Asia, Europe and North and South America were analyzed. The median age (interquartile range [IQR]) for reaching death or permanent ventilation was ~ 7.3 (5.9-10.5) months. The median age (IQR) at permanent ventilation was ~ 12.7 (6.9-16.4) months and at death was ~ 41.2 (7.3-not applicable) months. No patients were able to sit without support or achieved any level of crawling, standing or walking.
Interpretation: Findings from ANCHOVY were consistent with published natural history data on Type 1 SMA demonstrating the disease's devastating course, which markedly differed from risdiplam-treated infants (FIREFISH Part 2). The results provide meaningful additions to the literature, including a broader geographical representation.
Keywords: ANCHOVY; FIREFISH; SMA natural history; Spinal muscular atrophy; Type 1 SMA.
© 2022. The Author(s).
Conflict of interest statement
CC is a site principal investigator for Biogen and F. Hoffmann-La Roche Ltd clinical trials, and has received advisory fees from Novartis TG. VD and GPC have no conflicts of interest. RM has received fees from Biogen, F. Hoffmann-La Roche Ltd and Novartis Gene Therapies. MMB is a site principal investigator for Biogen and F. Hoffmann-La Roche Ltd clinical trials, and has received honoraria for advisory boards and speaker's fees from Biogen, F. Hoffmann-La Roche Ltd, and Novartis. KS is a site principal investigator for Biogen and Novartis Gene Therapies clinical trials, has received honoraria for advisory boards from Biogen, Novartis, and Roche/Chugai and speaker’s fees from Biogen and Novartis. IG and JH are employees of F. Hoffmann-La Roche Ltd. AD, MEK, KG and RSS are employees of, and hold shares in, F. Hoffmann-La Roche Ltd. BTD has received grants from Biogen, CureSMA, F. Hoffmann-La Roche Ltd, Fibrogen, Ionis Pharmaceuticals, U.S. National Institutes of Health/National Institute of Neurological Disorders and Stroke, PTC Therapeutics, Sarepta Pharmaceuticals, Slaney Family Fund for SMA, Spinal Muscular Atrophy Foundation, Summit and Working on Walking Fund; and is a board member for Amicus Inc., AveXis, Biogen, F. Hoffmann-La Roche Ltd grants, Genentech, Sarepta Pharmaceuticals and Vertex.
Figures
Similar articles
-
Safety and efficacy of risdiplam in patients with type 1 spinal muscular atrophy (FIREFISH part 2): secondary analyses from an open-label trial.Lancet Neurol. 2022 Dec;21(12):1110-1119. doi: 10.1016/S1474-4422(22)00339-8. Epub 2022 Oct 14. Lancet Neurol. 2022. PMID: 36244364 Clinical Trial.
-
Long-Term Comparative Efficacy and Safety of Risdiplam and Nusinersen in Children with Type 1 Spinal Muscular Atrophy.Adv Ther. 2024 Jun;41(6):2414-2434. doi: 10.1007/s12325-024-02845-6. Epub 2024 May 5. Adv Ther. 2024. PMID: 38705943 Free PMC article.
-
Risdiplam-Treated Infants with Type 1 Spinal Muscular Atrophy versus Historical Controls.N Engl J Med. 2021 Jul 29;385(5):427-435. doi: 10.1056/NEJMoa2102047. N Engl J Med. 2021. PMID: 34320287
-
Risdiplam: A Review in Spinal Muscular Atrophy.CNS Drugs. 2022 Apr;36(4):401-410. doi: 10.1007/s40263-022-00910-8. Epub 2022 Mar 13. CNS Drugs. 2022. PMID: 35284988 Review.
-
How does risdiplam compare with other treatments for Types 1-3 spinal muscular atrophy: a systematic literature review and indirect treatment comparison.J Comp Eff Res. 2022 Apr;11(5):347-370. doi: 10.2217/cer-2021-0216. Epub 2022 Jan 18. J Comp Eff Res. 2022. PMID: 35040693 Review.
Cited by
-
Respiratory morbidity in patients with spinal muscular atrophy-a changing world in the light of disease-modifying therapies.Front Pediatr. 2024 Mar 14;12:1366943. doi: 10.3389/fped.2024.1366943. eCollection 2024. Front Pediatr. 2024. PMID: 38550632 Free PMC article. Review.
-
Clinical and Functional Characteristics of a New Phenotype of SMA Type I among a National Sample of Spanish Children: A Cross-Sectional Study.Children (Basel). 2023 May 16;10(5):892. doi: 10.3390/children10050892. Children (Basel). 2023. PMID: 37238440 Free PMC article.
-
Identifying Biomarkers of Spinal Muscular Atrophy for Further Development.J Neuromuscul Dis. 2023;10(5):937-954. doi: 10.3233/JND-230054. J Neuromuscul Dis. 2023. PMID: 37458045 Free PMC article.
-
Motor and neurocognitive profiles of children with symptomatic spinal muscular atrophy type 1 with two copies of SMN2 before and after treatment: a longitudinal observational study.Front Neurol. 2024 Feb 21;15:1326528. doi: 10.3389/fneur.2024.1326528. eCollection 2024. Front Neurol. 2024. PMID: 38450080 Free PMC article.
References
-
- Darras B, Monani U, De Vivo D. Genetic disorders affecting the motor neuron: spinal muscular atrophy. In: Swaiman K, Ashwal S, Ferriero D, Schor N, Finkel R, Gropman A, editors. Swaiman’s pediatric neurology: principles and practice. 6. Elsevier; 2017. pp. 1057–1064.
Publication types
MeSH terms
Associated data
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical

