A long-acting C-natriuretic peptide for achondroplasia
- PMID: 35858423
- PMCID: PMC9335275
- DOI: 10.1073/pnas.2201067119
A long-acting C-natriuretic peptide for achondroplasia
Abstract
The C-natriuretic peptide (CNP) analog vosoritide has recently been approved for treatment of achondroplasia in children. However, the regimen requires daily subcutaneous injections in pediatric patients over multiple years. The present work sought to develop a long-acting CNP that would provide efficacy equal to or greater than that of vosoritide but require less frequent injections. We used a technology for half-life extension, whereby a drug is attached to tetra-polyethylene glycol hydrogels (tetra-PEG) by β-eliminative linkers that cleave at predetermined rates. These hydrogels-fabricated as uniform ∼60-μm microspheres-are injected subcutaneously, where they serve as a stationary depot to slowly release the drug into the systemic circulation. We prepared a highly active, stable CNP analog-[Gln6,14]CNP-38-composed of the 38 C-terminal amino acids of human CNP-53 containing Asn to Gln substitutions to preclude degradative deamidation. Two microsphere [Gln6,14]CNP-38 conjugates were prepared, with release rates designed to allow once-weekly and once-monthly administration. After subcutaneous injection of the conjugates in mice, [Gln6,14]CNP-38 was slowly released into the systemic circulation and showed biphasic elimination pharmacokinetics with terminal half-lives of ∼200 and ∼600 h. Both preparations increased growth of mice comparable to or exceeding that produced by daily vosoritide. Simulations of the pharmacokinetics in humans indicated that plasma [Gln6,14]CNP-38 levels should be maintained within a therapeutic window over weekly, biweekly, and likely, monthly dosing intervals. Compared with vosoritide, which requires ∼30 injections per month, microsphere [Gln6,14]CNP-38 conjugates-especially the biweekly and monthly dosing-could provide an alternative that would be well accepted by physicians, patients, and patient caregivers.
Keywords: C-natriuretic peptide; achondroplasia; half-life extension; hydrogel microsphere.
Conflict of interest statement
Competing interest statement: All authors are employees and hold options or stock in ProLynx.
Figures


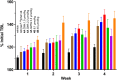
 ) and vosoritide (•). (B) Effects of [Gln6,14]CNP-38 and vosoritide on TBL (nasoanal + tail length) growth of 3-wk-old FVB/nJ male mice. Groups of 3-wk-old wild-type mice (n = 6) were given daily subcutaneous administrations of 70 nmol/kg CNP variants or vehicle for 5 wk. cGMP, cyclic guanosine monophosphate. ns, not significant.
) and vosoritide (•). (B) Effects of [Gln6,14]CNP-38 and vosoritide on TBL (nasoanal + tail length) growth of 3-wk-old FVB/nJ male mice. Groups of 3-wk-old wild-type mice (n = 6) were given daily subcutaneous administrations of 70 nmol/kg CNP variants or vehicle for 5 wk. cGMP, cyclic guanosine monophosphate. ns, not significant.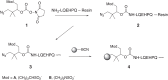
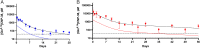
 ), and assuming dose linearity the simulated dose adjustment to 20 nmol to keep the peptide ∼40 pM (
), and assuming dose linearity the simulated dose adjustment to 20 nmol to keep the peptide ∼40 pM ( ) for 1 wk. (B) Single dose of 4B containing 700 nmol [Gln6,14]CNP-38/mouse (
) for 1 wk. (B) Single dose of 4B containing 700 nmol [Gln6,14]CNP-38/mouse ( ) and simulated dose-adjustment to 85 nmol to maintain the released peptide ≥40 pM (
) and simulated dose-adjustment to 85 nmol to maintain the released peptide ≥40 pM ( ) for 1 mo. Data points are mean ± SD.
) for 1 mo. Data points are mean ± SD.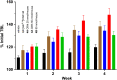
Similar articles
-
TransCon CNP, a Sustained-Release C-Type Natriuretic Peptide Prodrug, a Potentially Safe and Efficacious New Therapeutic Modality for the Treatment of Comorbidities Associated with Fibroblast Growth Factor Receptor 3-Related Skeletal Dysplasias.J Pharmacol Exp Ther. 2019 Sep;370(3):459-471. doi: 10.1124/jpet.119.258251. Epub 2019 Jun 24. J Pharmacol Exp Ther. 2019. PMID: 31235532
-
Pharmacokinetics and Exposure-Response of Vosoritide in Children with Achondroplasia.Clin Pharmacokinet. 2022 Feb;61(2):263-280. doi: 10.1007/s40262-021-01059-1. Epub 2021 Aug 25. Clin Pharmacokinet. 2022. PMID: 34431071 Free PMC article. Clinical Trial.
-
Efficacy of vosoritide in the treatment of achondroplasia.Drugs Today (Barc). 2022 Sep;58(9):451-456. doi: 10.1358/dot.2022.58.9.3422313. Drugs Today (Barc). 2022. PMID: 36102905
-
Vosoritide: First Approval.Drugs. 2021 Nov;81(17):2057-2062. doi: 10.1007/s40265-021-01623-w. Drugs. 2021. PMID: 34694597 Review.
-
[Genetic basis for skeletal disease. CNP therapy for achondroplasia].Clin Calcium. 2010 Aug;20(8):1212-8. Clin Calcium. 2010. PMID: 20675932 Review. Japanese.
Cited by
-
The Treatment of Growth Disorders in Childhood and Adolescence.Dtsch Arztebl Int. 2024 Feb 9;121(3):96-106. doi: 10.3238/arztebl.m2023.0247. Dtsch Arztebl Int. 2024. PMID: 38051162 Free PMC article. Review.
-
C-type natriuretic peptide/cGMP/FoxO3 signaling attenuates hyperproliferation of pericytes from patients with pulmonary arterial hypertension.Commun Biol. 2024 Jun 6;7(1):693. doi: 10.1038/s42003-024-06375-3. Commun Biol. 2024. PMID: 38844781 Free PMC article.
References
-
- Legeai-Mallet L., Savarirayan R., Novel therapeutic approaches for the treatment of achondroplasia. Bone 141, 115579 (2020). - PubMed
-
- Prickett T. C., A Espiner E., Circulating products of C-type natriuretic peptide and links with organ function in health and disease. Peptides 132, 170363 (2020). - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

