Generalized Arterial Calcification of Infancy (GACI): Optimizing Care with a Multidisciplinary Approach
- PMID: 35677616
- PMCID: PMC9167688
- DOI: 10.2147/JMDH.S251861
Generalized Arterial Calcification of Infancy (GACI): Optimizing Care with a Multidisciplinary Approach
Abstract
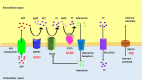
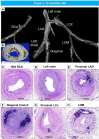
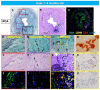
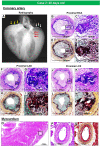
It is very unusual to see evidence of arterial calcification in infants and children, and when detected, genetic disorders of calcium metabolism should be suspected. Generalized arterial calcification of infancy (GACI) is a hereditary disease, which is characterized by severe arterial calcification of medium sized arteries, mostly involving the media with marked intimal proliferation and ectopic mineralization of the extravascular tissues. It is caused by inactivating variants in genes encoding either ENPP1, in a majority of cases (70-75%), or ABCC6, in a minority (9-10%). Despite similar histologic appearances between ENPP1 and ABCC6 deficiencies, including arterial calcification, organ calcification, and cardiovascular calcification, mortality is higher in subjects carrying the ENPP1 versus ABCC6 variants (40% vs 10%, respectively). Overall mortality in individuals with GACI is high (55%) before the age of 6 months, with 24.4% dying in utero or being stillborn. Rare cases show spontaneous regression with age, while others who survive into adulthood often manifest musculoskeletal complications (osteoarthritis and interosseous membrane ossification), enthesis mineralization, and cervical spine fusion. Despite recent advances in the understanding of the genetic mechanisms underlying this disease, there is still no ideal therapy for the resolution of vascular calcification in GACI. Although bisphosphonates with anti-calcification properties have been commonly used for the treatment of CAGI, their benefit is controversial, with favorable results reported at one year and questionable benefit with delayed initiation of treatment. Enzyme replacement therapy with administration of recombinant form of ENPP1 prevents calcification and mortality, improves hypertension and cardiac function, and prevents intimal proliferation and osteomalacia in mouse models of ENPP1 deficiency. Therefore, newer treatments targeting genes are on the horizon. In this article, we review up to date knowledge of the understanding of GACI, its clinical, pathologic, and etiologic understanding and treatment in support of more comprehensive care of GACI patients.
Keywords: ATP binding cassette subfamily C member 6; ectonucleotide pyrophosphatase/phosphodiesterase 1; generalized arterial calcification of infancy; vascular calcification.
© 2022 Kawai et al.
Conflict of interest statement
Dr. Aloke V. Finn received consultant fees/honoraria from Abbott Vascular, Amgen, Biosensors, Boston Scientific, Celonova, Cook Medical, CSI, Lutonix Bard, Sinomed, and Terumo Corporation. Dr. Renu Virmani is a consultant of Abbott Vascular, Boston Scientific, Celonova, OrbusNeich Medical, Terumo Corporation, W. L. Gore, Edwards Lifesciences, Cook Medical, CSI, ReCor Medical, SinoMedical Sciences Technology, Surmodics, Bard BD and a scientific Advisory Board Member of Medtronic and Xeltis. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or any other conflict with the subject matter or materials discussed.
Figures

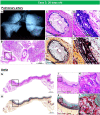
Similar articles
-
Ectopic Calcification and Hypophosphatemic Rickets: Natural History of ENPP1 and ABCC6 Deficiencies.J Bone Miner Res. 2021 Nov;36(11):2193-2202. doi: 10.1002/jbmr.4418. Epub 2021 Aug 16. J Bone Miner Res. 2021. PMID: 34355424 Free PMC article.
-
ENPP1 enzyme replacement therapy improves blood pressure and cardiovascular function in a mouse model of generalized arterial calcification of infancy.Dis Model Mech. 2018 Oct 8;11(10):dmm035691. doi: 10.1242/dmm.035691. Dis Model Mech. 2018. PMID: 30158213 Free PMC article.
-
Case report: A rare homozygous variation in the ENPP1 gene, presenting with generalized arterial calcification of infancy in a Chinese infant.Front Cardiovasc Med. 2023 Mar 3;10:1105381. doi: 10.3389/fcvm.2023.1105381. eCollection 2023. Front Cardiovasc Med. 2023. PMID: 36937905 Free PMC article.
-
Generalized Arterial Calcification of Infancy (GACI).Arch Pediatr. 2024 Sep;31(4S1):4S21-4S26. doi: 10.1016/S0929-693X(24)00153-2. Arch Pediatr. 2024. PMID: 39343469 Review.
-
Autosomal recessive hypophosphatemic rickets type 2 due to ENPP1 deficiency (ARHR2).Arch Pediatr. 2024 Sep;31(4S1):4S27-4S32. doi: 10.1016/S0929-693X(24)00154-4. Arch Pediatr. 2024. PMID: 39343470 Review.
Cited by
-
Influence of DNA Methylation on Vascular Smooth Muscle Cell Phenotypic Switching.Int J Mol Sci. 2024 Mar 8;25(6):3136. doi: 10.3390/ijms25063136. Int J Mol Sci. 2024. PMID: 38542110 Free PMC article. Review.
-
Preterm Infant with Generalized Arterial Calcification of Infancy Who Survived Due to Early Diagnosis and Appropriate Treatment with Bisphosphonates: A Case Report.Children (Basel). 2024 Sep 27;11(10):1176. doi: 10.3390/children11101176. Children (Basel). 2024. PMID: 39457141 Free PMC article.
-
Inhibition of Vascular Smooth Muscle Cell Proliferation by ENPP1: The Role of CD73 and the Adenosine Signaling Axis.Cells. 2024 Jun 29;13(13):1128. doi: 10.3390/cells13131128. Cells. 2024. PMID: 38994980 Free PMC article.
-
Clinical and Molecular Characterization of a Patient with Generalized Arterial Calcification of Infancy Caused by Rare ABCC6 Mutation.J Pers Med. 2023 Dec 30;14(1):54. doi: 10.3390/jpm14010054. J Pers Med. 2023. PMID: 38248755 Free PMC article.
-
Homozygous splice-site variant in ENPP1 underlies generalized arterial calcification of infancy.BMC Pediatr. 2024 Nov 13;24(1):733. doi: 10.1186/s12887-024-05123-0. BMC Pediatr. 2024. PMID: 39538190 Free PMC article.
References
-
- Erbel R, Möhlenkamp S, Moebus S, et al. Coronary risk stratification, discrimination, and reclassification improvement based on quantification of subclinical coronary atherosclerosis: the Heinz Nixdorf Recall study. J Am Coll Cardiol. 2010;56(17):1397–1406. doi:10.1016/j.jacc.2010.06.030 - DOI - PubMed
-
- Taylor AJ, Bindeman J, Feuerstein I, Cao F, Brazaitis M, O’Malley PG. Coronary calcium independently predicts incident premature coronary heart disease over measured cardiovascular risk factors: mean three-year outcomes in the Prospective Army Coronary Calcium (PACC) project. J Am Coll Cardiol. 2005;46(5):807–814. doi:10.1016/j.jacc.2005.05.049 - DOI - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

