Sensitizing tumors to anti-PD-1 therapy by promoting NK and CD8+ T cells via pharmacological activation of FOXO3
- PMID: 34887262
- PMCID: PMC8663085
- DOI: 10.1136/jitc-2021-002772
Sensitizing tumors to anti-PD-1 therapy by promoting NK and CD8+ T cells via pharmacological activation of FOXO3
Abstract
Background: Stimulating antitumor immunity by blocking programmed death-1 (PD-1) or its ligand (programmed death-ligand 1 (PD-L1) is a promising antitumor therapy. However, numerous patients respond poorly to PD-1/PD-L1 blockade. Unresponsiveness to immune-checkpoint blockade (ICB) can cast significant challenges to the therapeutic options for patients with hard-to-treat tumors. There is an unmet clinical need to establish new therapeutic approaches for mitigating ICB unresponsiveness in patients. In this study, we investigated the efficacy and role of low-dose antineoplastic agent SN-38 or metformin in sensitizing unresponsive tumors to respond to ICB therapy.
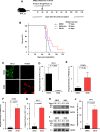
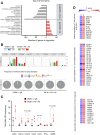
Methods: We assessed the significant pathological relationships between PD-L1 and FOXO3 expression and between PD-L1 and c-Myc or STAT3 expression in patients with various tumors. We determined the efficacy of low-dose SN-38 or metformin in sensitizing unresponsive tumors to respond to anti-PD-1 therapy in a syngeneic tumor system. We deciphered novel therapeutic mechanisms underlying the SN-38 and anti-PD-1 therapy-mediated engagement of natural killer (NK) or CD8+ T cells to infiltrate tumors and boost antitumor immunity.
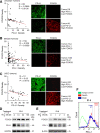
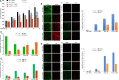
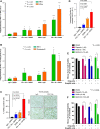
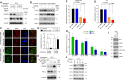
Results: We showed that PD-L1 protein level was inversely associated with FOXO3 protein level in patients with ovarian, breast, and hepatocellular tumors. Low-dose SN-38 or metformin abrogated PD-L1 protein expression, promoted FOXO3 protein level, and significantly increased the animal survival rate in syngeneic mouse tumor models. SN-38 or metformin sensitized unresponsive tumors responding to anti-PD-1 therapy by engaging NK or CD8+ T cells to infiltrate the tumor microenvironment (TME) and secret interferon-γ and granzyme B to kill tumors. SN-38 suppressed the levels of c-Myc and STAT3 proteins, which controlled PD-L1 expression. FOXO3 was essential for SN38-mediated PD-L1 suppression. The expression of PD-L1 was compellingly linked to that of c-Myc or STAT3 in patients with the indicated tumors.
Conclusion: We show that SN-38 or metformin can boost antitumor immunity in the TME by inhibiting c-Myc and STAT3 through FOXO3 activation. These results may provide novel insight into ameliorating patient response to overarching immunotherapy for tumors.
Keywords: CD8-positive T-lymphocytes; combined modality therapy; immunomodulation; immunotherapy; natural killer T-cells.
© Author(s) (or their employer(s)) 2021. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ.
Conflict of interest statement
Competing interests: None declared.
Figures


Similar articles
-
Enhanced Efficacy of Simultaneous PD-1 and PD-L1 Immune Checkpoint Blockade in High-Grade Serous Ovarian Cancer.Cancer Res. 2021 Jan 1;81(1):158-173. doi: 10.1158/0008-5472.CAN-20-1674. Epub 2020 Nov 6. Cancer Res. 2021. PMID: 33158814 Free PMC article.
-
Preferential Expression of Programmed Death Ligand 1 Protein in Tumor-Associated Macrophages and Its Potential Role in Immunotherapy for Hepatocellular Carcinoma.Int J Mol Sci. 2021 Apr 29;22(9):4710. doi: 10.3390/ijms22094710. Int J Mol Sci. 2021. PMID: 33946835 Free PMC article.
-
Releasing the brakes of tumor immunity with anti-PD-L1 and pushing its accelerator with L19-IL2 cures poorly immunogenic tumors when combined with radiotherapy.J Immunother Cancer. 2021 Mar;9(3):e001764. doi: 10.1136/jitc-2020-001764. J Immunother Cancer. 2021. PMID: 33688020 Free PMC article.
-
Augmenting Anticancer Immunity Through Combined Targeting of Angiogenic and PD-1/PD-L1 Pathways: Challenges and Opportunities.Front Immunol. 2020 Nov 5;11:598877. doi: 10.3389/fimmu.2020.598877. eCollection 2020. Front Immunol. 2020. PMID: 33250900 Free PMC article. Review.
-
Programmed cell death protein-1 (PD-1)/programmed death-ligand-1 (PD-L1) axis in hepatocellular carcinoma: prognostic and therapeutic perspectives.Clin Transl Oncol. 2019 Jun;21(6):702-712. doi: 10.1007/s12094-018-1975-4. Epub 2018 Nov 1. Clin Transl Oncol. 2019. PMID: 30387047 Review.
Cited by
-
Glycolysis in tumor microenvironment as a target to improve cancer immunotherapy.Front Cell Dev Biol. 2022 Sep 19;10:1013885. doi: 10.3389/fcell.2022.1013885. eCollection 2022. Front Cell Dev Biol. 2022. PMID: 36200045 Free PMC article. Review.
-
Metformin enhances the antitumor activity of oncolytic herpes simplex virus HF10 (canerpaturev) in a pancreatic cell cancer subcutaneous model.Sci Rep. 2022 Dec 13;12(1):21570. doi: 10.1038/s41598-022-25065-w. Sci Rep. 2022. PMID: 36513720 Free PMC article.
-
Tislelizumab plus cetuximab and irinotecan in refractory microsatellite stable and RAS wild-type metastatic colorectal cancer: a single-arm phase 2 study.Nat Commun. 2024 Aug 23;15(1):7255. doi: 10.1038/s41467-024-51536-x. Nat Commun. 2024. PMID: 39179622 Free PMC article. Clinical Trial.
-
Modulating Glycolysis to Improve Cancer Therapy.Int J Mol Sci. 2023 Jan 30;24(3):2606. doi: 10.3390/ijms24032606. Int J Mol Sci. 2023. PMID: 36768924 Free PMC article. Review.
-
New Achievements from Molecular Biology and Treatment Options for Refractory/Relapsed Ovarian Cancer-A Systematic Review.Cancers (Basel). 2023 Nov 10;15(22):5356. doi: 10.3390/cancers15225356. Cancers (Basel). 2023. PMID: 38001616 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
