Mechanisms of Genome Instability in the Fragile X-Related Disorders
- PMID: 34681027
- PMCID: PMC8536109
- DOI: 10.3390/genes12101633
Mechanisms of Genome Instability in the Fragile X-Related Disorders
Abstract
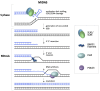
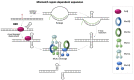
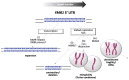
The Fragile X-related disorders (FXDs), which include the intellectual disability fragile X syndrome (FXS), are disorders caused by expansion of a CGG-repeat tract in the 5' UTR of the X-linked FMR1 gene. These disorders are named for FRAXA, the folate-sensitive fragile site that localizes with the CGG-repeat in individuals with FXS. Two pathological FMR1 allele size classes are distinguished. Premutation (PM) alleles have 54-200 repeats and confer the risk of fragile X-associated tremor/ataxia syndrome (FXTAS) and fragile X-associated primary ovarian insufficiency (FXPOI). PM alleles are prone to both somatic and germline expansion, with female PM carriers being at risk of having a child with >200+ repeats. Inheritance of such full mutation (FM) alleles causes FXS. Contractions of PM and FM alleles can also occur. As a result, many carriers are mosaic for different sized alleles, with the clinical presentation depending on the proportions of these alleles in affected tissues. Furthermore, it has become apparent that the chromosomal fragility of FXS individuals reflects an underlying problem that can lead to chromosomal numerical and structural abnormalities. Thus, large numbers of CGG-repeats in the FMR1 gene predisposes individuals to multiple forms of genome instability. This review will discuss our current understanding of these processes.
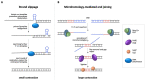
Keywords: aneuploidy; base excision repair (BER); break induced replication (BIR); chromosome fragility; microhomology mediated end-joining (MMEJ); mitotic DNA synthesis (MiDAS); repeat contractions; repeat expansion; repeat mosaicism.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Unstable mutations in the FMR1 gene and the phenotypes.Adv Exp Med Biol. 2012;769:78-114. doi: 10.1007/978-1-4614-5434-2_6. Adv Exp Med Biol. 2012. PMID: 23560306 Free PMC article. Review.
-
Both cis and trans-acting genetic factors drive somatic instability in female carriers of the FMR1 premutation.Sci Rep. 2022 Jun 21;12(1):10419. doi: 10.1038/s41598-022-14183-0. Sci Rep. 2022. PMID: 35729184 Free PMC article.
-
Presence of inclusions positive for polyglycine containing protein, FMRpolyG, indicates that repeat-associated non-AUG translation plays a role in fragile X-associated primary ovarian insufficiency.Hum Reprod. 2016 Jan;31(1):158-68. doi: 10.1093/humrep/dev280. Epub 2015 Nov 3. Hum Reprod. 2016. PMID: 26537920 Free PMC article.
-
[FMR1 PREMUTATION CARRIERS - ARE THEY REALLY ASYMPTOMATIC?].Harefuah. 2018 Apr;157(4):241-244. Harefuah. 2018. PMID: 29688643 Review. Hebrew.
-
High functioning male with fragile X syndrome and fragile X-associated tremor/ataxia syndrome.Am J Med Genet A. 2015 Sep;167A(9):2154-61. doi: 10.1002/ajmg.a.37125. Epub 2015 Apr 29. Am J Med Genet A. 2015. PMID: 25920745
Cited by
-
Fragile X Syndrome Caused by Maternal Somatic Mosaicism of FMR1 Gene: Case Report and Literature Review.Genes (Basel). 2022 Sep 8;13(9):1609. doi: 10.3390/genes13091609. Genes (Basel). 2022. PMID: 36140775 Free PMC article. Review.
-
Searching for New Z-DNA/Z-RNA Binding Proteins Based on Structural Similarity to Experimentally Validated Zα Domain.Int J Mol Sci. 2022 Jan 11;23(2):768. doi: 10.3390/ijms23020768. Int J Mol Sci. 2022. PMID: 35054954 Free PMC article.
-
Somatic and intergenerational G4C2 hexanucleotide repeat instability in a human C9orf72 knock-in mouse model.Nucleic Acids Res. 2024 Jun 10;52(10):5732-5755. doi: 10.1093/nar/gkae250. Nucleic Acids Res. 2024. PMID: 38597682 Free PMC article.
-
Editorial for the Fragile X Syndrome Genetics Special Issue: May 2023.Genes (Basel). 2023 May 25;14(6):1148. doi: 10.3390/genes14061148. Genes (Basel). 2023. PMID: 37372328 Free PMC article.
-
Dynamic alternative DNA structures in biology and disease.Nat Rev Genet. 2023 Apr;24(4):211-234. doi: 10.1038/s41576-022-00539-9. Epub 2022 Oct 31. Nat Rev Genet. 2023. PMID: 36316397 Free PMC article. Review.
References
-
- Fu Y.H., Kuhl D.P., Pizzuti A., Pieretti M., Sutcliffe J.S., Richards S., Verkerk A.J., Holden J.J., Fenwick R.G., Jr., Warren S.T., et al. Variation of the CGG repeat at the fragile X site results in genetic instability: Resolution of the Sherman paradox. Cell. 1991;67:1047–1058. doi: 10.1016/0092-8674(91)90283-5. - DOI - PubMed
-
- Verkerk A.J., Pieretti M., Sutcliffe J.S., Fu Y.H., Kuhl D.P., Pizzuti A., Reiner O., Richards S., Victoria M.F., Zhang F.P., et al. Identification of a gene (FMR-1) containing a CGG repeat coincident with a breakpoint cluster region exhibiting length variation in fragile X syndrome. Cell. 1991;65:905–914. doi: 10.1016/0092-8674(91)90397-H. - DOI - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical
Research Materials

