"Don, doff, discard" to "don, doff, decontaminate"-FFR and mask integrity and inactivation of a SARS-CoV-2 surrogate and a norovirus following multiple vaporised hydrogen peroxide-, ultraviolet germicidal irradiation-, and dry heat decontaminations
- PMID: 34010337
- PMCID: PMC8133425
- DOI: 10.1371/journal.pone.0251872
"Don, doff, discard" to "don, doff, decontaminate"-FFR and mask integrity and inactivation of a SARS-CoV-2 surrogate and a norovirus following multiple vaporised hydrogen peroxide-, ultraviolet germicidal irradiation-, and dry heat decontaminations
Abstract
Background: As the SARS-CoV-2 pandemic accelerates, the supply of personal protective equipment remains under strain. To combat shortages, re-use of surgical masks and filtering facepiece respirators has been recommended. Prior decontamination is paramount to the re-use of these typically single-use only items and, without compromising their integrity, must guarantee inactivation of SARS-CoV-2 and other contaminating pathogens.
Aim: We provide information on the effect of time-dependent passive decontamination (infectivity loss over time during room temperature storage in a breathable bag) and evaluate inactivation of a SARS-CoV-2 surrogate and a non-enveloped model virus as well as mask and respirator integrity following active multiple-cycle vaporised hydrogen peroxide (VHP), ultraviolet germicidal irradiation (UVGI), and dry heat (DH) decontamination.
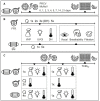
Methods: Masks and respirators, inoculated with infectious porcine respiratory coronavirus or murine norovirus, were submitted to passive decontamination or single or multiple active decontamination cycles; viruses were recovered from sample materials and viral titres were measured via TCID50 assay. In parallel, filtration efficiency tests and breathability tests were performed according to EN standard 14683 and NIOSH regulations.
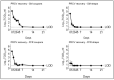
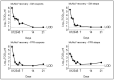
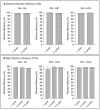
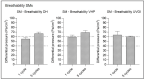
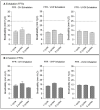
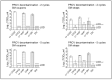
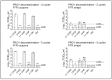
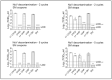
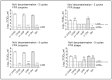
Results and discussion: Infectious porcine respiratory coronavirus and murine norovirus remained detectable on masks and respirators up to five and seven days of passive decontamination. Single and multiple cycles of VHP-, UVGI-, and DH were shown to not adversely affect bacterial filtration efficiency of masks. Single- and multiple UVGI did not adversely affect respirator filtration efficiency, while VHP and DH induced a decrease in filtration efficiency after one or three decontamination cycles. Multiple cycles of VHP-, UVGI-, and DH slightly decreased airflow resistance of masks but did not adversely affect respirator breathability. VHP and UVGI efficiently inactivated both viruses after five, DH after three, decontamination cycles, permitting demonstration of a loss of infectivity by more than three orders of magnitude. This multi-disciplinal approach provides important information on how often a given PPE item may be safely reused.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
Similar articles
-
The use of germicidal ultraviolet light, vaporized hydrogen peroxide and dry heat to decontaminate face masks and filtering respirators contaminated with a SARS-CoV-2 surrogate virus.J Hosp Infect. 2020 Nov;106(3):577-584. doi: 10.1016/j.jhin.2020.08.025. Epub 2020 Sep 1. J Hosp Infect. 2020. PMID: 32889029 Free PMC article.
-
The use of germicidal ultraviolet light, vaporised hydrogen peroxide and dry heat to decontaminate face masks and filtering respirators contaminated with an infectious norovirus.Infect Prev Pract. 2021 Mar;3(1):100111. doi: 10.1016/j.infpip.2020.100111. Epub 2020 Dec 28. Infect Prev Pract. 2021. PMID: 34316573 Free PMC article.
-
Addressing personal protective equipment (PPE) decontamination: Methylene blue and light inactivates severe acute respiratory coronavirus virus 2 (SARS-CoV-2) on N95 respirators and medical masks with maintenance of integrity and fit.Infect Control Hosp Epidemiol. 2022 Jul;43(7):876-885. doi: 10.1017/ice.2021.230. Epub 2021 May 21. Infect Control Hosp Epidemiol. 2022. PMID: 34016200 Free PMC article.
-
Decontamination of respirators amid shortages due to SARS-CoV-2.Photochem Photobiol Sci. 2021 Jul;20(7):955-965. doi: 10.1007/s43630-021-00064-4. Epub 2021 Jun 12. Photochem Photobiol Sci. 2021. PMID: 34118013 Free PMC article. Review.
-
A Practical Approach to Filtering Facepiece Respirator Decontamination and Reuse: Ultraviolet Germicidal Irradiation.Curr Treat Options Infect Dis. 2021;13(2):35-46. doi: 10.1007/s40506-021-00247-8. Epub 2021 Apr 6. Curr Treat Options Infect Dis. 2021. PMID: 33841050 Free PMC article. Review.
Cited by
-
Aerosolized Hydrogen Peroxide Decontamination of N95 Respirators, with Fit-Testing and Viral Inactivation, Demonstrates Feasibility for Reuse during the COVID-19 Pandemic.mSphere. 2022 Oct 26;7(5):e0030322. doi: 10.1128/msphere.00303-22. Epub 2022 Aug 30. mSphere. 2022. PMID: 36040048 Free PMC article.
-
aPDI meets PPE: photochemical decontamination in healthcare using methylene blue-where are we now, where will we go?Photochem Photobiol Sci. 2024 Feb;23(2):215-223. doi: 10.1007/s43630-023-00514-1. Epub 2024 Jan 2. Photochem Photobiol Sci. 2024. PMID: 38165604
-
Communications enhance sustainable intentions despite other ongoing crises.Sustain Sci. 2024;19(6):1997-2012. doi: 10.1007/s11625-024-01556-9. Epub 2024 Sep 14. Sustain Sci. 2024. PMID: 39526227 Free PMC article.
-
Effect of five decontamination methods on face masks and filtering facepiece respirators contaminated with Staphylococcus aureus and Pseudomonas aeruginosa.Access Microbiol. 2022 Mar 30;4(3):000342. doi: 10.1099/acmi.0.000342. eCollection 2022. Access Microbiol. 2022. PMID: 35693470 Free PMC article.
-
Bioaerosol Inactivation by a Cold Plasma Ionizer Coupled with an Electrostatic Precipitator.Microorganisms. 2024 Sep 21;12(9):1923. doi: 10.3390/microorganisms12091923. Microorganisms. 2024. PMID: 39338597 Free PMC article.
References
-
- World Health Organization (WHO). Rational use of personal protective equipment for coronavirus disease 2019 (COVID-19). Who. 2020;2019: 1–7.
-
- WHO. Strengthening the health system response to COVID-19 Recommendations for the WHO European Region Policy brief. 2020; 8. https://www.thelancet.com/journals/langlo/article/PIIS2214-109X
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

