SARS-CoV-2 viral load dynamics and real-time RT-PCR cycle threshold interpretation in symptomatic non-hospitalised individuals in New Zealand: a multicentre cross sectional observational study
- PMID: 33838922
- PMCID: PMC7980174
- DOI: 10.1016/j.pathol.2021.01.007
SARS-CoV-2 viral load dynamics and real-time RT-PCR cycle threshold interpretation in symptomatic non-hospitalised individuals in New Zealand: a multicentre cross sectional observational study
Abstract
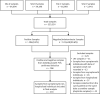
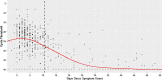
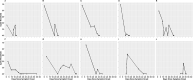
We conducted a multicentre cross sectional observational study of laboratory, public health and hospitalisation data for PCR-confirmed COVID-19 cases within the New Zealand Northern Region, between 12 February and 8 June 2020. The aim of this study was to describe population level SARS-CoV-2 upper respiratory tract (URT) viral load dynamics by stratifying positivity rates and polymerase chain reaction (PCR) cycle threshold (Ct) values of URT samples from COVID-19 cases by days since symptom onset, and to explore utility of Ct values in determining length of time post-infection and thus potential infectivity. Of 123,124 samples tested for SARS-CoV-2 by PCR, 579 samples (407 positive and 172 negative) from 368 symptomatic non-hospitalised individuals with PCR-confirmed infection were included. Sample positivity rate was 61.5% (8/13) for pre-symptomatic samples, rising to 93.2% (317/340) for samples collected during the purported symptomatic infectious period (days 0-10 post-symptom onset), and dropping to 36.3% (82/226) for post-infectious period samples (day 11 onwards). URT viral load peaked shortly after symptom onset, with median Ct values ranging 20.00-29.99 until 15 days post-symptom onset, and >30.00 after this time. Of samples with a Ct value of <20.00, 96.1% were collected during the symptomatic infectious period. However, of samples with a Ct value ≥30.00 and ≥35.00, 46.9% and 18.5%, respectively, were also collected during the symptomatic infectious period. The findings of this study indicate that at or soon after symptom onset represents the optimum time to test for SARS-CoV-2 in the URT, with median Ct values suggesting the useful testing window extends until around 15 days post-symptom onset. In asymptomatic individuals or those with unknown dates of symptom onset, Ct values <20.00 imply recent onset/potential infectivity, but Ct values ≥30.00 or ≥35.00 do not exclude recent onset/potential infectivity. Individual sample Ct values should not be used as an absolute marker of length of time post-infection or to exclude infectivity where date of symptom onset is unavailable.
Keywords: COVID-19; New Zealand; PCR; SARS-CoV-2; cycle threshold.
Copyright © 2021 Royal College of Pathologists of Australasia. Published by Elsevier B.V. All rights reserved.
Figures
Similar articles
-
Investigation of Viral Load Cycle Threshold Values in Patients with SARS-CoV-2 Associated Pneumonia with Real-Time PCR Method.J Infect Dev Ctries. 2021 Oct 31;15(10):1408-1414. doi: 10.3855/jidc.14281. J Infect Dev Ctries. 2021. PMID: 34780363
-
Infectivity assay for detection of SARS-CoV-2 in samples from patients with COVID-19.J Med Virol. 2021 Oct;93(10):5917-5923. doi: 10.1002/jmv.27145. Epub 2021 Jun 29. J Med Virol. 2021. PMID: 34139026 Free PMC article.
-
Cycle threshold values in RT-PCR to determine dynamics of SARS-CoV-2 viral load: An approach to reduce the isolation period for COVID-19 patients.J Med Virol. 2021 Dec;93(12):6794-6797. doi: 10.1002/jmv.27206. Epub 2021 Jul 23. J Med Virol. 2021. PMID: 34264527 Free PMC article.
-
Assessing severe acute respiratory syndrome coronavirus 2 infectivity by reverse-transcription polymerase chain reaction: A systematic review and meta-analysis.Rev Med Virol. 2022 Sep;32(5):e2342. doi: 10.1002/rmv.2342. Epub 2022 Apr 2. Rev Med Virol. 2022. PMID: 35366033 Free PMC article. Review.
-
Preanalytical Issues and Cycle Threshold Values in SARS-CoV-2 Real-Time RT-PCR Testing: Should Test Results Include These?ACS Omega. 2021 Mar 6;6(10):6528-6536. doi: 10.1021/acsomega.1c00166. eCollection 2021 Mar 16. ACS Omega. 2021. PMID: 33748564 Free PMC article. Review.
Cited by
-
Cycle Threshold Values as Indication of Increasing SARS-CoV-2 New Variants, England, 2020-2022.Emerg Infect Dis. 2023 Oct;29(10):2024-2031. doi: 10.3201/eid2910.230030. Epub 2023 Sep 7. Emerg Infect Dis. 2023. PMID: 37678158 Free PMC article.
-
SARS-CoV-2 Variants of Concern: Presumptive Identification via Sanger Sequencing Analysis of the Receptor Binding Domain (RBD) Region of the S Gene.Diagnostics (Basel). 2023 Mar 27;13(7):1256. doi: 10.3390/diagnostics13071256. Diagnostics (Basel). 2023. PMID: 37046474 Free PMC article.
-
Approach to hematopoietic cell transplant candidates with respiratory viral detection.Front Pediatr. 2024 Jan 18;11:1339239. doi: 10.3389/fped.2023.1339239. eCollection 2023. Front Pediatr. 2024. PMID: 38304442 Free PMC article. Review.
-
The Deceptive COVID-19: Lessons from Common Molecular Diagnostics and a Novel Plan for the Prevention of the Next Pandemic.Diseases. 2023 Jan 28;11(1):20. doi: 10.3390/diseases11010020. Diseases. 2023. PMID: 36810534 Free PMC article.
-
COVID or no COVID: Interpreting inconclusive SARS-CoV-2 qPCR results in different populations and platforms.J Clin Virol Plus. 2023 Jun;3(2):100145. doi: 10.1016/j.jcvp.2023.100145. Epub 2023 Mar 9. J Clin Virol Plus. 2023. PMID: 36941981 Free PMC article.
References
-
- Baker M., Kvalsvig A., Verrall A.J., et al. New Zealand's elimination strategy for the COVID-19 pandemic and what is required to make it work. N Z Med J. 2020;133:10–14. - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

