Preclinical Characterization of the Distribution, Catabolism, and Elimination of a Polatuzumab Vedotin-Piiq (POLIVY®) Antibody-Drug Conjugate in Sprague Dawley Rats
- PMID: 33806916
- PMCID: PMC8004598
- DOI: 10.3390/jcm10061323
Preclinical Characterization of the Distribution, Catabolism, and Elimination of a Polatuzumab Vedotin-Piiq (POLIVY®) Antibody-Drug Conjugate in Sprague Dawley Rats
Abstract
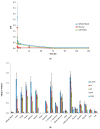
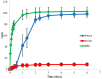
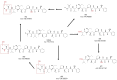
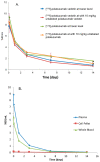
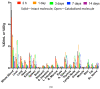
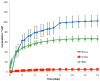
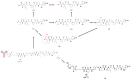
Polatuzumab vedotin (or POLIVY®), an antibody-drug conjugate (ADC) composed of a polatuzumab monoclonal antibody conjugated to monomethyl auristatin E (MMAE) via a cleavable dipeptide linker, has been approved by the United States Food and Drug Administration (FDA) for the treatment of diffuse large B-cell lymphoma (DLBCL). To support the clinical development of polatuzumab vedotin, we characterized the distribution, catabolism/metabolism, and elimination properties of polatuzumab vedotin and its unconjugated MMAE payload in Sprague Dawley rats. Several radiolabeled probes were developed to track the fate of different components of the ADC, with 125I and 111In used to label the antibody component and 3H to label the MMAE payload of the ADC. Following a single intravenous administration of the radiolabeled probes into normal or bile-duct cannulated rats, blood, various tissues, and excreta samples were collected over 7-14 days post-dose and analyzed for radioactivity and to characterize the metabolites/catabolites. The plasma radioactivity of polatuzumab vedotin showed a biphasic elimination profile similar to that of unconjugated polatuzumab but different from unconjugated radiolabeled MMAE, which had a fast clearance. The vast majority of the radiolabeled MMAE in plasma remained associated with antibodies, with a minor fraction as free MMAE and MMAE-containing catabolites. Similar to unconjugated mAb, polatuzumab vedotin showed a nonspecific distribution to multiple highly perfused organs, including the lungs, heart, liver, spleen, and kidneys, where the ADC underwent catabolism to release MMAE and other MMAE-containing catabolites. Both polatuzumab vedotin and unconjugated MMAE were mainly eliminated through the biliary fecal route (>90%) and a small fraction (<10%) was eliminated through renal excretion in the form of catabolites/metabolites, among which, MMAE was identified as the major species, along with several other minor species. These studies provided significant insight into ADC's absorption, distribution, metabolism, and elimination (ADME) properties, which supports the clinical development of POLIVY.
Keywords: antibody–drug conjugate (ADC); distribution, catabolism, and elimination (DME); mass balance; monomethyl auristatin E (MMAE); polatuzumab vedotin (POLIVY).
Conflict of interest statement
The authors are Genentech employees at the time of conducting these studies and declare no conflict of interest.
Figures
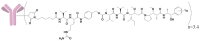


Similar articles
-
Monomethyl auristatin E (MMAE), a payload for multiple antibody drug conjugates (ADCs), demonstrates differential red blood cell partitioning across human and animal species.Xenobiotica. 2024 Aug;54(8):511-520. doi: 10.1080/00498254.2024.2345849. Epub 2024 Sep 27. Xenobiotica. 2024. PMID: 38647387
-
Polatuzumab Vedotin: First Global Approval.Drugs. 2019 Sep;79(13):1467-1475. doi: 10.1007/s40265-019-01175-0. Drugs. 2019. PMID: 31352604 Free PMC article. Review.
-
Evaluation and use of an anti-cynomolgus monkey CD79b surrogate antibody-drug conjugate to enable clinical development of polatuzumab vedotin.Br J Pharmacol. 2019 Oct;176(19):3805-3818. doi: 10.1111/bph.14784. Epub 2019 Aug 24. Br J Pharmacol. 2019. PMID: 31270798 Free PMC article.
-
Polatuzumab vedotin pharmacokinetics in a hemodialysis patient with diffuse large B-cell lymphoma.Cancer Chemother Pharmacol. 2024 Mar;93(3):265-268. doi: 10.1007/s00280-023-04593-w. Epub 2023 Sep 26. Cancer Chemother Pharmacol. 2024. PMID: 37750932
-
Polatuzumab vedotin: an investigational anti-CD79b antibody drug conjugate for the treatment of diffuse large B-cell lymphoma.Expert Opin Investig Drugs. 2020 Oct;29(10):1079-1088. doi: 10.1080/13543784.2020.1800638. Epub 2020 Aug 12. Expert Opin Investig Drugs. 2020. PMID: 32700972 Review.
Cited by
-
Modular Smart Molecules for PSMA-Targeted Chemotherapy.Mol Cancer Ther. 2022 Nov 3;21(11):1701-1709. doi: 10.1158/1535-7163.MCT-22-0160. Mol Cancer Ther. 2022. PMID: 35999662 Free PMC article.
-
A novel approach to quantitate biodistribution and transduction of adeno-associated virus gene therapy using radiolabeled AAV vectors in mice.Mol Ther Methods Clin Dev. 2024 Aug 19;32(3):101326. doi: 10.1016/j.omtm.2024.101326. eCollection 2024 Sep 12. Mol Ther Methods Clin Dev. 2024. PMID: 39286334 Free PMC article.
-
Payload-Binding Fab Fragments Increase the Therapeutic Index of MMAE Antibody-Drug Conjugates.Mol Cancer Ther. 2023 Apr 3;22(4):459-470. doi: 10.1158/1535-7163.MCT-22-0440. Mol Cancer Ther. 2023. PMID: 36723609 Free PMC article.
-
Immune-related adverse events of antibody-based biological medicines in cancer therapy.J Cell Mol Med. 2024 Jul;28(13):e18470. doi: 10.1111/jcmm.18470. J Cell Mol Med. 2024. PMID: 38963257 Free PMC article. Review.
-
Monitoring In Vivo Performances of Protein-Drug Conjugates Using Site-Selective Dual Radiolabeling and Ex Vivo Digital Imaging.J Med Chem. 2022 May 12;65(9):6953-6968. doi: 10.1021/acs.jmedchem.2c00401. Epub 2022 May 2. J Med Chem. 2022. PMID: 35500280 Free PMC article.
References
-
- Li D., Lee D., Dere R.C., Zheng B., Yu S.F., Fuh F.K., Kozak K.R., Chung S., Bumbaca Yadav D., Nazzal D., et al. Evaluation and use of an anti-cynomolgus monkey CD79b surrogate antibody-drug conjugate to enable clinical development of polatuzumab vedotin. Br. J. Pharmacol. 2019;176:3805–3818. doi: 10.1111/bph.14784. - DOI - PMC - PubMed
-
- Adcetris, FDA CDER (Center for Drug Evaluation and Research), Clinical Pharmacology and Biopharmaceutics Review. Application Number 125399Orig1s000. [(accessed on 23 March 2016)];2011 Available online: http://www.accessdata.fda.gov/drugsatfda_docs/label/2011/125388s000,1253....
LinkOut - more resources
Full Text Sources
Other Literature Sources

