Therapeutic Assessment of Combination Therapy with a Neprilysin Inhibitor and Angiotensin Type 1 Receptor Antagonist on Angiotensin II-Induced Atherosclerosis, Abdominal Aortic Aneurysms, and Hypertension
- PMID: 33707301
- PMCID: PMC8140395
- DOI: 10.1124/jpet.121.000525
Therapeutic Assessment of Combination Therapy with a Neprilysin Inhibitor and Angiotensin Type 1 Receptor Antagonist on Angiotensin II-Induced Atherosclerosis, Abdominal Aortic Aneurysms, and Hypertension
Erratum in
-
Correction to "Therapeutic Assessment of Combination Therapy with a Neprilysin Inhibitor and Angiotensin Type 1 Receptor Antagonist on Angiotensin II - Induced Atherosclerosis, Abdominal Aortic Aneurysms, and Hypertensin".J Pharmacol Exp Ther. 2021 Sep;378(3):322. doi: 10.1124/jpet.121.000525err. J Pharmacol Exp Ther. 2021. PMID: 34561274 No abstract available.
Abstract
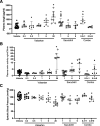
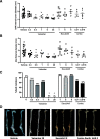
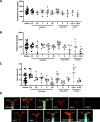
Combined neprilysin (NEP) inhibition (sacubitril) and angiotensin type 1 receptor (AT1R) antagonism (valsartan) is used in the treatment of congestive heart failure and is gaining interest for other angiotensin II (AngII)-related cardiovascular diseases. In addition to heart failure, AngII promotes hypertension, atherosclerosis, and abdominal aortic aneurysms (AAAs). Similarly, NEP substrates or products have broad effects on the cardiovascular system. In this study, we examined NEP inhibition (with sacubitril) and AT1R antagonism (with valsartan) alone or in combination on AngII-induced hypertension, atherosclerosis, or AAAs in male low-density lipoprotein receptor-deficient mice. Preliminary studies assessed drug delivery via osmotic minipumps for simultaneous release of sacubitril and/or valsartan with AngII over 28 days. Mice were infused with AngII (1000 ng/kg per minute) in the absence (vehicle) or presence of sacubitril (1, 6, or 9 mg/kg per day), valsartan (0.3, 0.5, 1, 6, or 20 mg/kg per day), or the combination thereof (1 and 0.3, or 9 or 0.5 mg/kg per day of sacubitril and valsartan, respectively). Plasma AngII and renin concentrations increased 4-fold at higher valsartan doses, indicative of removal of AngII negative feedback on renin. Sacubitril doubled plasma AngII concentrations at lower doses (1 mg/kg per day). Valsartan dose-dependently decreased systolic blood pressure, aortic atherosclerosis, and AAAs of AngII-infused mice, whereas sacubitril had no effect on atherosclerosis or AAAs but reduced blood pressure of AngII-infused mice. Combination therapy with sacubitril and valsartan did not provide additive benefits. These results suggest limited effects of combination therapy with NEP inhibition and AT1R antagonism against AngII-induced hypertension, atherosclerosis, or AAAs. SIGNIFICANCE STATEMENT: The combination of valsartan (angiotensin type 1 receptor antagonist) and sacubitril (neprilysin inhibitor) did not provide benefit above valsartan alone on AngII-induced hypertension, atherosclerosis, or abdominal aortic aneurysms in low-density lipoprotein receptor-deficient male mice. These results do not support this drug combination in therapy of these AngII-induced cardiovascular diseases.
Copyright © 2021 The Author(s).
Conflict of interest statement
No author has an actual or perceived conflict of interest with the contents of this article.
Figures

Similar articles
-
Insulinotropic Effects of Neprilysin and/or Angiotensin Receptor Inhibition in Mice.Front Endocrinol (Lausanne). 2022 Jun 6;13:888867. doi: 10.3389/fendo.2022.888867. eCollection 2022. Front Endocrinol (Lausanne). 2022. PMID: 35733766 Free PMC article.
-
PD123319 augments angiotensin II-induced abdominal aortic aneurysms through an AT2 receptor-independent mechanism.PLoS One. 2013 Apr 12;8(4):e61849. doi: 10.1371/journal.pone.0061849. Print 2013. PLoS One. 2013. PMID: 23593499 Free PMC article.
-
Sacubitril/valsartan: A novel angiotensin receptor-neprilysin inhibitor.Indian Heart J. 2018 Jul;70 Suppl 1(Suppl 1):S102-S110. doi: 10.1016/j.ihj.2018.01.002. Epub 2018 Jan 8. Indian Heart J. 2018. PMID: 30122239 Free PMC article. Review.
-
Sacubitril/valsartan (LCZ696) significantly reduces aldosterone and increases cGMP circulating levels in a canine model of RAAS activation.Eur J Pharm Sci. 2019 Feb 1;128:103-111. doi: 10.1016/j.ejps.2018.11.037. Epub 2018 Nov 30. Eur J Pharm Sci. 2019. PMID: 30508581
-
The Sacubitril/Valsartan, a First-in-Class, Angiotensin Receptor Neprilysin Inhibitor (ARNI): Potential Uses in Hypertension, Heart Failure, and Beyond.Curr Cardiol Rep. 2018 Jan 27;20(1):5. doi: 10.1007/s11886-018-0944-4. Curr Cardiol Rep. 2018. PMID: 29374807 Review.
Cited by
-
Angiotensin Receptor-Neprilysin Inhibitor (ARNI) and Cardiac Arrhythmias.Int J Mol Sci. 2021 Aug 20;22(16):8994. doi: 10.3390/ijms22168994. Int J Mol Sci. 2021. PMID: 34445698 Free PMC article. Review.
-
Twenty Years of Studying AngII (Angiotensin II)-Induced Abdominal Aortic Pathologies in Mice: Continuing Questions and Challenges to Provide Insight Into the Human Disease.Arterioscler Thromb Vasc Biol. 2022 Mar;42(3):277-288. doi: 10.1161/ATVBAHA.121.317058. Epub 2022 Jan 20. Arterioscler Thromb Vasc Biol. 2022. PMID: 35045728 Free PMC article. Review.
References
-
- Abassi ZA, Tate JE, Golomb E, Keiser HR (1992) Role of neutral endopeptidase in the metabolism of endothelin. Hypertension 20:89–95. - PubMed
-
- Acosta S, Gottsäter A, Engström G, Melander O, Zarrouk M, Nilsson PM, Smith JG (2018) B-type natriuretic peptide for prediction of incident clinically significant abdominal aortic aneurysm: a population-based prospective study. Vasc Med 23:46–51. - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources

