Clinical Management of Anemia in Patients with Myelodysplastic Syndromes: An Update on Emerging Therapeutic Options
- PMID: 33531837
- PMCID: PMC7846829
- DOI: 10.2147/CMAR.S240600
Clinical Management of Anemia in Patients with Myelodysplastic Syndromes: An Update on Emerging Therapeutic Options
Abstract
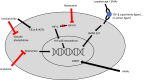
For the majority of patients with lower-risk myelodysplastic syndrome (LR-MDS), one of the primary clinical goals is to alleviate the symptoms associated with the resultant cytopenias and to minimize the transfusion burden. While supportive red blood cell (RBC) transfusions and erythropoiesis-stimulating agents (ESAs) may lead to clinical improvement, frequent transfusions are often complicated by iron overload and decreased quality of life; furthermore, most patients either do not respond to ESAs or will eventually develop resistance. As such, there is a great need for further therapeutic options in the management of anemia related to MDS. Several additional therapeutics are now available in select patients with LR-MDS and symptomatic anemia including luspatercept, lenalidomide, and immunosuppressive therapy. Furthermore, several novel agents are currently in development to address this area of clinical need such as imetelstat and roxadustat. In this article, we review the currently available therapeutic options for symptomatic anemia in LR-MDS as well as review the therapeutic agents in development.
Keywords: clinical trials; erythropoiesis-stimulating agents; myelodysplastic syndrome; novel agents.
© 2021 Lewis et al.
Conflict of interest statement
A.M.Z. received research funding (institutional) from Celgene/BMS, AbbVie, Astex, Pfizer, Medimmune/AstraZeneca, Boehringer-Ingelheim, Trovagene/Cardiff oncology, Incyte, Takeda, Novartis, Aprea, and ADC Therapeutics. A.M.Z participated in advisory boards, and/or had a consultancy with and received honoraria from AbbVie, Otsuka, Pfizer, Celgene/BMS, Jazz, Incyte, Agios, Boehringer-Ingelheim, Novartis, Acceleron, Astellas, Daiichi Sankyo, Cardinal Health, Taiho, Seattle Genetics, BeyondSpring, Trovagene/Cardiff Oncology, Takeda, Ionis, Amgen, Janssen, Epizyme, Syndax, and Tyme. A.M.Z served on clinical trial committees for Novartis, AbbVie, Geron and Celgene/BMS. A.M.Z received travel support for meetings from Pfizer, Novartis, and Trovagene/Cardiff Oncology. None of these relationships were related to the development of this manuscript. R.L. and J.P.B. have no conflicts of interest.
Figures
Similar articles
-
Treatment options for lower-risk myelodysplastic syndromes. Where are we now?Ther Adv Hematol. 2021 Jan 14;12:2040620720986641. doi: 10.1177/2040620720986641. eCollection 2021. Ther Adv Hematol. 2021. PMID: 33505645 Free PMC article. Review.
-
New Approaches to Myelodysplastic Syndrome Treatment.Curr Treat Options Oncol. 2022 May;23(5):668-687. doi: 10.1007/s11864-022-00965-1. Epub 2022 Mar 23. Curr Treat Options Oncol. 2022. PMID: 35320468 Review.
-
Luspatercept in low-risk myelodysplastic syndromes: a paradigm shift in treatment strategies.Expert Opin Biol Ther. 2024 Apr;24(4):233-241. doi: 10.1080/14712598.2024.2336086. Epub 2024 Mar 30. Expert Opin Biol Ther. 2024. PMID: 38555469 Review.
-
Raising the bar for lower-risk myelodysplastic syndromes.Leuk Lymphoma. 2023 Jun;64(6):1082-1091. doi: 10.1080/10428194.2023.2197536. Epub 2023 Apr 8. Leuk Lymphoma. 2023. PMID: 37029589 Review.
-
Treatment of Anemia in Transfusion-Dependent and Non-Transfusion-Dependent Lower-Risk MDS: Current and Emerging Strategies.Hemasphere. 2019 Oct 30;3(6):e314. doi: 10.1097/HS9.0000000000000314. eCollection 2019 Dec. Hemasphere. 2019. PMID: 31976486 Free PMC article. Review.
Cited by
-
Risk-Adapted, Individualized Treatment Strategies of Myelodysplastic Syndromes (MDS) and Chronic Myelomonocytic Leukemia (CMML).Cancers (Basel). 2021 Mar 31;13(7):1610. doi: 10.3390/cancers13071610. Cancers (Basel). 2021. PMID: 33807279 Free PMC article. Review.
-
Testosterone therapy as a novel approach to the management of cytopenias in myelodysplastic neoplasms: a review of literature and case report.J Cancer Res Clin Oncol. 2024 Aug 29;150(8):404. doi: 10.1007/s00432-024-05844-w. J Cancer Res Clin Oncol. 2024. PMID: 39207555 Free PMC article. Review.
-
Efficacy and safety of lenalidomide in diffuse large B-cell lymphoma: a meta-analysis of randomized controlled trials.Clin Exp Med. 2023 Aug;23(4):1161-1169. doi: 10.1007/s10238-022-00920-2. Epub 2022 Oct 31. Clin Exp Med. 2023. PMID: 36315313 Free PMC article. Review.
-
Clinical Pharmacokinetics and Pharmacodynamics of Roxadustat.Clin Pharmacokinet. 2022 Mar;61(3):347-362. doi: 10.1007/s40262-021-01095-x. Epub 2021 Dec 14. Clin Pharmacokinet. 2022. PMID: 34905154 Free PMC article. Review.
References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

