Inebilizumab: First Approval
- PMID: 32729016
- PMCID: PMC7387876
- DOI: 10.1007/s40265-020-01370-4
Inebilizumab: First Approval
Abstract
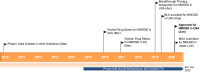
Inebilizumab (Uplizna™; inebilizumab-cdon in the USA) is a humanised anti-CD19 monoclonal antibody being developed by Viela Bio for the treatment of a range of autoimmune diseases associated with CD19-expressing B cells. Inebilizumab targets and depletes CD19-expressing B cells through antibody-dependent cell-mediated cytotoxicity. In June 2020, inebilizumab received its first global approval in the USA for the treatment of neuromyelitis optica spectrum disorder (NMOSD) in adult patients who are seropositive for immunoglobulin G autoantibodies against aquaporin-4 (AQP4-IgG). The drug is also undergoing clinical evaluation for kidney transplant desensitization, myasthenia gravis, and IgG4-related disease. This article summarizes the milestones in the development of inebilizumab leading to this first approval for the treatment of AQP4-IgG seropositive NMOSD.
Conflict of interest statement
During the peer review process the manufacturer of the agent under review was offered an opportunity to comment on the article. Changes resulting from any comments received were made by the author on the basis of scientific completeness and accuracy. James E. Frampton is a salaried employee of Adis International Ltd/Springer Nature, is responsible for the article content and declares no relevant conflicts of interest.
Figures
Similar articles
-
Inebilizumab-cdon: USFDA Approved for the Treatment of NMOSD (Neuromyelitis Optica Spectrum Disorder).Curr Drug Discov Technol. 2022;19(1):e140122193419. doi: 10.2174/1570163818666210519103001. Curr Drug Discov Technol. 2022. PMID: 34011260
-
Inebilizumab: A Review in Neuromyelitis Optica Spectrum Disorder.CNS Drugs. 2022 Oct;36(10):1133-1141. doi: 10.1007/s40263-022-00949-7. Epub 2022 Sep 7. CNS Drugs. 2022. PMID: 36070074 Free PMC article. Review.
-
[Efficacy and Safety of Inebilizumab, an Anti-CD19 Monoclonal Antibody, for the Treatment of Neuromyelitis Optica Spectrum Disorder: Based on the N-MOmentum Trial].Brain Nerve. 2024 Oct;76(10):1153-1160. doi: 10.11477/mf.1416202751. Brain Nerve. 2024. PMID: 39370840 Review. Japanese.
-
AQP4-IgG-seronegative patient outcomes in the N-MOmentum trial of inebilizumab in neuromyelitis optica spectrum disorder.Mult Scler Relat Disord. 2022 Jan;57:103356. doi: 10.1016/j.msard.2021.103356. Epub 2021 Nov 1. Mult Scler Relat Disord. 2022. PMID: 35158465 Clinical Trial.
-
Safety and efficacy of inebilizumab for the treatment of neuromyelitis optica spectrum disorder: end-of-study results from the open-label period of the N-MOmentum trial.Lancet Neurol. 2024 Jun;23(6):588-602. doi: 10.1016/S1474-4422(24)00077-2. Lancet Neurol. 2024. PMID: 38760098 Clinical Trial.
Cited by
-
Pediatric Neuromyelitis Optica Spectrum Disorder: Case Series and Literature Review.Life (Basel). 2021 Dec 23;12(1):19. doi: 10.3390/life12010019. Life (Basel). 2021. PMID: 35054412 Free PMC article. Review.
-
Neuromyelitis Optica Spectrum Disorder Management in the Setting of Chronic Hepatitis B and Latent Tuberculosis: A Case Report.Neurohospitalist. 2023 Oct;13(4):361-363. doi: 10.1177/19418744231171464. Epub 2023 May 4. Neurohospitalist. 2023. PMID: 37701252 Free PMC article.
-
New Targeted Agents in Myasthenia Gravis and Future Therapeutic Strategies.J Clin Med. 2022 Oct 28;11(21):6394. doi: 10.3390/jcm11216394. J Clin Med. 2022. PMID: 36362622 Free PMC article. Review.
-
Treatment of Neuromyelitis Optica Spectrum Disorders.Int J Mol Sci. 2021 Aug 11;22(16):8638. doi: 10.3390/ijms22168638. Int J Mol Sci. 2021. PMID: 34445343 Free PMC article. Review.
-
Neuromyelitis Optica Spectrum Disorder Treatment-Current and Future Prospects.Int J Mol Sci. 2021 Mar 10;22(6):2801. doi: 10.3390/ijms22062801. Int J Mol Sci. 2021. PMID: 33802046 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources